Impact Factor
Theranostics 2022; 12(2):734-746. doi:10.7150/thno.66468 This issue Cite
Research Paper
Ion drugs for precise orthotopic tumor management by in situ the generation of toxic ion and drug pools
State Key Laboratory of Molecular Vaccinology and Molecular Diagnostics and Center for Molecular Imaging and Translational Medicine, School of Public Health, Xiamen University, Xiamen 361102, China.
Abstract
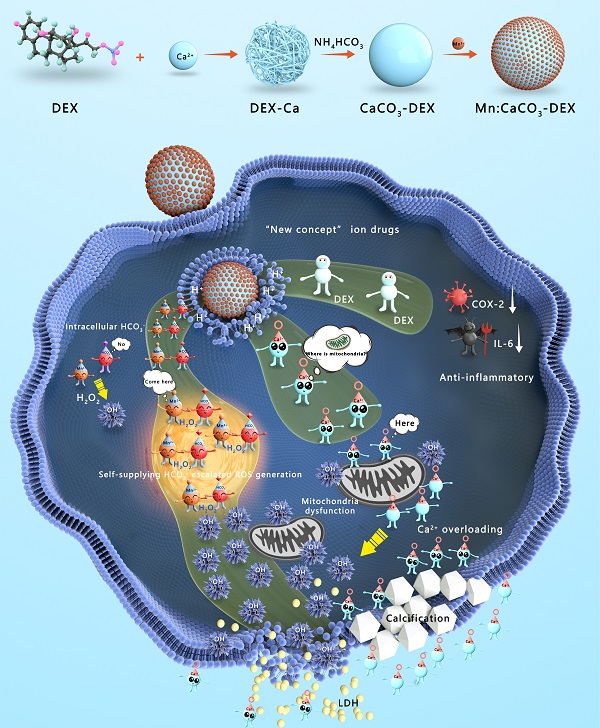
Background: Asymmetric intracellular and extracellular ionic gradients are critical to the survivability of mammalian cells. Given the importance of manganese (Mn2+), calcium (Ca2+), and bicarbonate (HCO3-) ions, any alteration of the ion-content balance could induce a series of cellular responses. HCO3- plays an indispensable role for Mn-mediated Fenton-like reaction, but this is difficult to achieve because bicarbonates are tightly regulated by live cells, and are limited in anticancer efficacy.
Methods: A responsive and biodegradable biomineral, Mn-doped calcium carbonate integrated with dexamethasone phosphate (DEX) (Mn:CaCO3-DEX), was reported to enable synergistic amplification of tumor oxidative stress, reduce inflammation, and induce Ca-overload cell apoptosis by elevating the intracellular and extracellular ionic gradients.
Results: Under the acidic environment in tumor region, the ions (Mn2+, CO32-, Ca2+) were released by the degradation of Mn:CaCO3-DEX and then escalated oxidative stresses by triggering a HCO3--indispensable Mn-based Fenton-like reaction and breaking Ca2+ ion homeostasis to cause oxidative stress in cells and calcification. The released anti-inflammatory and antitumor drug, DEX, could alleviate the inflammatory environment. The investigations in vitro and in vivo demonstrated that the synergistic oncotherapy could effectively inhibit the growth of subcutaneous tumors and orthotopic liver tumors. Notably, normal cells showed greater tolerance of the synergistic influences.
Conclusion: As an ion drug, Mn:CaCO3-DEX is an excellent potential diagnostic agent for precise orthotopic tumor management by the generation in situ of toxic ion and drug pools in the environment of tumor region, with synergistic effects of enhanced chemodynamic therapy, calcification, and anti-inflammation effects.
Keywords: Tumor acidic environment, Internal ions overload, Chemodynamic therapy, Calcification, Anti-inflammatory
 Global reach, higher impact
Global reach, higher impact