13.3
Impact Factor
Theranostics 2022; 12(2):720-733. doi:10.7150/thno.65796 This issue Cite
Research Paper
Loss of function of GATA3 induces basal-like mammary tumors
1. Guangdong Provincial Key Laboratory of Regional Immunity and Diseases, International Cancer Center, Marshall Laboratory of Biomedical Engineering, Shenzhen University Health Science Center, Shenzhen 518060, China.
2. Department of Pathology, Shenzhen University Health Science Center, Shenzhen 518060, China.
3. Dewitt Daughtry Family Department of Surgery, Sylvester Comprehensive Cancer Center, University of Miami, Miami, FL 33136, USA.
4. Xinjiang Uigur Autonomous Region People's Hospital, Xinjiang, 830001, China.
5. Department of Biochemistry and Molecular Biology, International Cancer Center, Shenzhen University Health Science Center, Shenzhen 518060, China.
6. Department of Anatomy and Histology, Shenzhen University Health Science Center, Shenzhen 518060, China.
# These authors contributed equally to this article
Abstract
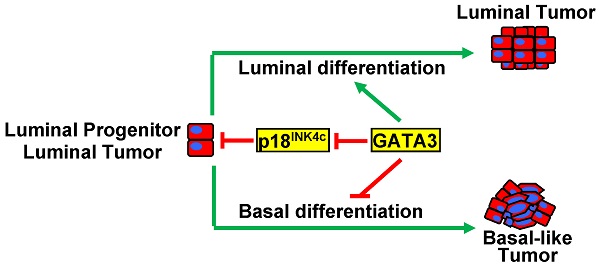
Purpose: GATA3 is a transcription factor essential for mammary luminal epithelial cell differentiation. Expression of GATA3 is absent or significantly reduced in basal-like breast cancers. Gata3 loss-of-function impairs cell proliferation, making it difficult to investigate the role of GATA3 deficiency in vivo. We previously demonstrated that CDK inhibitor p18INK4c (p18) is a downstream target of GATA3 and restrains mammary epithelial cell proliferation and tumorigenesis. Whether and how loss-of-function of GATA3 results in basal-like breast cancers remains elusive.
Methods: We generated mutant mouse strains with heterozygous germline deletion of Gata3 in p18 deficient backgrounds and developed a Gata3 depleted mammary tumor model system to determine the role of Gata3 loss in controlling cell proliferation and aberrant differentiation in mammary tumor development and progression.
Results: Haploid loss of Gata3 reduced mammary epithelial cell proliferation with induction of p18, impaired luminal differentiation, and promoted basal differentiation in mammary glands. p18 deficiency induced luminal type mammary tumors and rescued the proliferative defect caused by haploid loss of Gata3. Haploid loss of Gata3 accelerated p18 deficient mammary tumor development and changed the properties of these tumors, resulting in their malignant and luminal-to-basal transformation. Expression of Gata3 negatively correlated with basal differentiation markers in MMTV-PyMT mammary tumor cells. Depletion of Gata3 in luminal tumor cells also reduced cell proliferation with induction of p18 and promoted basal differentiation. We confirmed that expression of GATA3 and basal markers are inversely correlated in human basal-like breast cancers.
Conclusions: This study provides the first genetic evidence demonstrating that loss-of-function of GATA3 directly induces basal-like breast cancer. Our finding suggests that basal-like breast cancer may also originate from luminal type cancer.
Keywords: Gata3 loss, p18INK4c, mammary tumor, basal differentiation
 Global reach, higher impact
Global reach, higher impact