13.3
Impact Factor
Theranostics 2022; 12(2):675-688. doi:10.7150/thno.64874 This issue Cite
Research Paper
β-glucan-coupled superparamagnetic iron oxide nanoparticles induce trained immunity to protect mice against sepsis
1. State Key Laboratory of Pharmaceutical Biotechnology, Division of Immunology, Medical School, Nanjing University, Nanjing 210093, China.
2. State Key Laboratory of Trauma, Burns and Combined Injury, Research Institute of Surgery, Daping Hospital, The Army Medical University, Chongqing 400042, China.
3. Jiangsu Key Laboratory of Molecular Medicine, Nanjing 210093, China.
Abstract
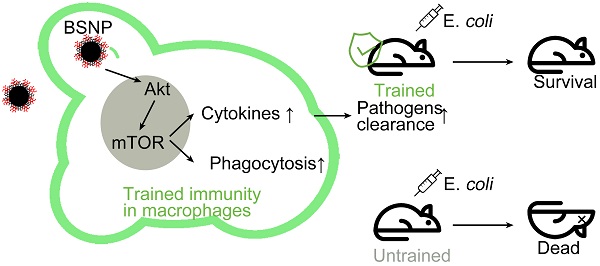
Background: Innate immune memory, also termed “trained immunity”, is thought to protect against experimental models of infection, including sepsis. Trained immunity via reprogramming monocytes/macrophages has been reported to result in enhanced inflammatory status and antimicrobial activity against infection in sepsis. However, a safe and efficient way to induce trained immunity remains unclear.
Methods: β-glucan is a prototypical agonist for inducing trained immunity. Ferumoxytol, superparamagnetic iron oxide (SPIO) with low cytotoxicity, has been approved by FDA for clinical use. We synthesized novel nanoparticles BSNPs by coupling β-glucan with SPIO. BSNPs were further conjugated with fluorescein for quantitative analysis and trace detection of β-glucan on BSNPs. Inflammatory cytokine levels were measured by ELISA and qRT-PCR, and the phagocytosis of macrophages was detected by flow cytometry and confocal microscopy. The therapeutic effect of BSNPs was evaluated on the well-established sepsis mouse model induced by both clinical Escherichia coli (E. coli) and cecal ligation and puncture (CLP).
Results: BSNPs were synthesized successfully with a 3:20 mass ratio of β-glucan and SPIO on BSNPs, which were mainly internalized by macrophages and accumulated in the lungs and livers of mice. BSNPs effectively reprogrammed macrophages to enhance the production of trained immunity markers and phagocytosis toward bacteria. BSNP-induced trained immunity protected mice against sepsis caused by E. coli and CLP and also against secondary infection. We found that BSNP treatment elevated Akt, S6, and 4EBP phosphorylation, while mTOR inhibitors decreased the trained immunity markers and phagocytosis enhanced by BSNPs. Furthermore, the PCR Array analysis revealed Igf1, Sesn1, Vegfa, and Rps6ka5 as possible key regulators of mTOR signaling during trained immunity. BSNP-induced trained immunity mainly regulated cellular signal transduction, protein modification, and cell cycle by modulating ATP binding and the kinase activity. Our results indicated that BSNPs induced trained immunity in an mTOR-dependent manner.
Conclusion: Our data highlight that the trained immunity of macrophages is an effective strategy against sepsis and suggest that BSNPs are a powerful tool for inducing trained immunity to prevent and treat sepsis and secondary infections.
Keywords: Sepsis, Macrophages, Trained immunity, β-glucan, SPIO
 Global reach, higher impact
Global reach, higher impact