13.3
Impact Factor
Theranostics 2022; 12(2):512-529. doi:10.7150/thno.64033 This issue Cite
Research Paper
Monocytes promote acute neuroinflammation and become pathological microglia in neonatal hypoxic-ischemic brain injury
1. Department of Neuroscience, Center for Brain Immunology and Glia (BIG), University of Virginia School of Medicine, Charlottesville, VA, USA.
2. Department of Anesthesiology, Taipei Veterans General Hospital and National Yang Ming Chiao Tung University School of Medicine, Taipei, Taiwan.
3. University of Louisville School of Medicine, Louisville, KY, USA.
4. St. Louis University School of Medicine, St. Louis, MO, USA.
5. Department of Anesthesiology, Rhode Island Hospital, Providence, RI, USA.
6. Emory University School of Medicine, Atlanta, GA, USA.
7. Emory Integrated Cellular Imaging, Atlanta, GA, USA.
8. Institute of BioPharmaceutical Sciences, National Sun Yat-sen University, Kaohsiung, Taiwan.
Abstract
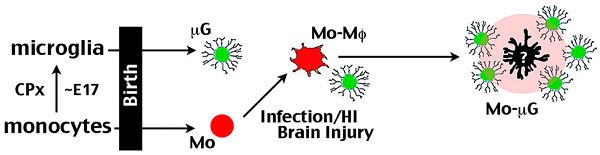
Rationale: Monocytes belong to the mononuclear phagocyte system and are immune responders to tissue injury and infection. There were also reports of monocytes transforming to microglia-like cells. Here we explore the roles of monocytes in microglia ontogeny and the pathogenesis of neonatal cerebral hypoxic-ischemic (HI) brain injury in mice.
Methods: We used three genetic methods to track the development of monocytes, including CX3CR1GFP/+; CCR2RFP/+ reporter mice, adoptive transfer of GFP+ monocytes, and fate-mapping with CCR2-CreER mice, in neonatal mouse brains with or without lipopolysaccharide (LPS, 0.3 mg/kg)-sensitized Vannucci HI. We also used genetic (CCR2RFP/ RFP, CCR2 knockout) and pharmacological methods (RS102895, a CCR2 antagonist) to test the roles of monocytic influx in LPS/HI brain injury.
Results: CCR2+ monocytes entered the late-embryonic brains via choroid plexus, but rapidly became CX3CR1+ amoeboid microglial cells (AMCs). The influx of CCR2+ monocytes declined after birth, but recurred after HI or LPS-sensitized HI (LPS/HI) brain injury, particularly in the hippocampus. The CCR2-CreER-based fate-mapping showed that CCR2+ monocytes became CD68+ TNFα+ macrophages within 4 d after LPS/HI, and maintained as TNFα+ MHCII+ macrophages or persisted as Tmem119+ Sall1+ P2RY12+ ramified microglia for at least five months after injury. Genetic deletion of the chemokine receptor CCR2 markedly diminished monocytic influx, the expression of pro- and anti-inflammatory cytokines, and brain damage. Post-LPS/HI application of RS102895 also reduced inflammatory responses and brain damage, leading to better cognitive functions.
Conclusion: These results suggest that monocytes promote acute inflammatory responses and may become pathological microglia long after the neonatal LPS/HI insult. Further, blocking the influx of monocytes may be a potential therapy for neonatal brain injury.
Keywords: CCR2, microglia, chorioamnionitis, hypoxic ischemic encephalopathy (HIE), monocyte-derived macrophages, neuroinflammation
 Global reach, higher impact
Global reach, higher impact