Impact Factor
Theranostics 2022; 12(1):76-86. doi:10.7150/thno.64759 This issue Cite
Research Paper
Smart NIR-II croconaine dye-peptide for enhanced photo-sonotheranostics of hepatocellular carcinoma
1. State Key Laboratory of Molecular Vaccinology and Molecular Diagnostics & Center for Molecular Imaging and Translational Medicine, School of Public Health, Xiamen University, Xiamen 361102, China.
2. International Institutes of Medicine, The Fourth Affiliated Hospital of Zhejiang University School of Medicine, Yiwu 322000, Zhejiang, China.
* S.L. and Y.Z. contributed equally to this study.
Abstract
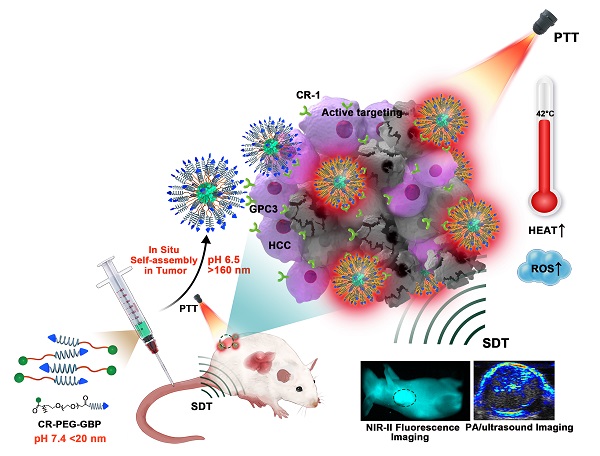
Background: Hepatocellular carcinoma (HCC) is associated with high morbidity and mortality rates. The development of novel nanomaterials represents an important direction for precise HCC theranostics. The combination of photothermal and sonodynamic therapy has provided great benefits for HCC therapy. Theranostic agents in the second near-infrared window (NIR-II, 1000-1700 nm) show great prospects because of their extraordinarily high detection sensitivity, resolution, and deep penetration.
Methods: A sharp pH-sensitive self-assembling Glypican-3 (GPC3)-binding peptide (GBP) dye, CR-PEG-GBP, was developed as an intelligent nanoprobe for NIR-II imaging and photoacoustic (PA) imaging-guided photothermal therapy (PTT) and sonodynamic therapy (SDT) of HCC.
Results: This small molecule assembled nanoprobe exhibited advantageous properties, such as responding to a decrease in pH (from normal tissue (pH 7.4) to the tumor microenvironment (pH ~6.5)) and aggregating - from small nanoprobes (<20 nm at pH 7.4) - into large nanoparticles (>160 nm at pH 6.5 and >510 nm at pH 5.5) that enables enhanced imaging and therapeutic effects. Because CR-PEG-GBP can self-aggregate in situ in an acidic tumor microenvironment, it shows high tumor accumulation and long tumor retention time, while being excretable from normal tissues and safe.
Conclusions: This intelligent self-assembling small molecule strategy provides a simple yet efficient solution for HCC theranostics and may open up new avenues for designing clinically translatable probes for HCC treatment.
Keywords: microenvironment, in situ self-assembly, theranostics, NIR-II, HCC
 Global reach, higher impact
Global reach, higher impact