13.3
Impact Factor
Theranostics 2021; 11(15):7527-7545. doi:10.7150/thno.54695 This issue Cite
Research Paper
Arginine starvation elicits chromatin leakage and cGAS-STING activation via epigenetic silencing of metabolic and DNA-repair genes
1. Institute of Biotechnology, National Tsing Hua University, Hsinchu 30013, Taiwan
2. Institute of Cellular and System Medicine, National Health Research Institutes, Miaoli County 35053, Taiwan
3. Institute of Molecular and Genomic Medicine, National Health Research Institutes, Miaoli County 35053, Taiwan
4. Metabolomics Core Laboratory, Healthy Aging Research Center, Chang Gung University, Taoyuan 33302, Taiwan
5. Graduate Institute of Biomedical Sciences, College of Medicine, Chang Gung University, Taoyuan 33302, Taiwan
6. Clinical Metabolomics Core Laboratory, Chang Gung Memorial Hospital at Linkou, Taoyuan 33305, Taiwan
7. TMU Research Center of Cancer Translational Medicine, Taipei Medical University, Taipei 110, Taiwan
8. CRISPR Gene Targeting Core Lab, Taipei Medical University, Taipei 110, Taiwan.
9. The PhD Program for Translational Medicine, College of Medical Science and Technology, Taipei Medical University, Taipei 110, Taiwan
10. The PhD Program for Cancer Molecular Biology and Drug Discovery, College of Medical Science and Technology, Taipei Medical University and Academia Sinica, Taipei 110, Taiwan
11. Department of Urology, School of Medicine, College of Medicine, Taipei Medical University, Taipei 110, Taiwan
12. Department of Urology, Taipei Medical University Hospital, Taipei 110, Taiwan
13. Department of Pathology, Chi Mei Medical Center, Tainan City 73657, Taiwan
14. Institute of Cancer Research, National Health Research Institutes, Miaoli County 35053, Taiwan
15. Institute of Medical Science and Technology, National Sun Yat-sen University, Kaohsiung 80424, Taiwan
16. Chinese Medicine Research Center and Institute of Integrated Medicine, China Medical University, Taichung 40402, Taiwan
17. Department of Biochemistry and Molecular Medicine, School of Medicine, University of California, Davis, Sacramento, CA 95817, USA
18. Department of Diabetes Complications and Metabolism, Beckman Research Institute, City of Hope, Duarte, CA 91010, USA
19. Institute of Medical Sciences, Tzu Chi University, Hualien 97004, Taiwan
20. Doctoral Degree Program in Translational Medicine, Tzu Chi University and Academia Sinica, Hualien 97004, Taiwan
21. Ph.D. Program for Cancer Biology and Drug Discovery, College of Medical Science and Technology, Taipei Medical University, Taipei 110, Taiwan
22. Department of Biochemistry and Molecular Medicine, Comprehensive Cancer Center, University of California, Davis, Sacramento, CA 95817, USA
Abstract
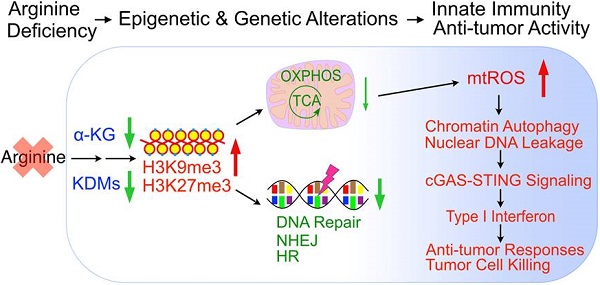
Rationale: One of the most common metabolic defects in cancers is the deficiency in arginine synthesis, which has been exploited therapeutically. Yet, challenges remain, and the mechanisms of arginine-starvation induced killing are largely unclear. Here, we sought to demonstrate the underlying mechanisms by which arginine starvation-induced cell death and to develop a dietary arginine-restriction xenograft model to study the in vivo effects.
Methods: Multiple castration-resistant prostate cancer cell lines were treated with arginine starvation followed by comprehensive analysis of microarray, RNA-seq and ChIP-seq were to identify the molecular and epigenetic pathways affected by arginine starvation. Metabolomics and Seahorse Flux analyses were used to determine the metabolic profiles. A dietary arginine-restriction xenograft mouse model was developed to assess the effects of arginine starvation on tumor growth and inflammatory responses.
Results: We showed that arginine starvation coordinately and epigenetically suppressed gene expressions, including those involved in oxidative phosphorylation and DNA repair, resulting in DNA damage, chromatin-leakage and cGAS-STING activation, accompanied by the upregulation of type I interferon response. We further demonstrated that arginine starvation-caused depletion of α-ketoglutarate and inactivation of histone demethylases are the underlying causes of epigenetic silencing. Significantly, our dietary arginine-restriction model showed that arginine starvation suppressed prostate cancer growth in vivo, with evidence of enhanced interferon responses and recruitment of immune cells.
Conclusions: Arginine-starvation induces tumor cell killing by metabolite depletion and epigenetic silencing of metabolic genes, leading to DNA damage and chromatin leakage. The resulting cGAS-STING activation may further enhance these killing effects.
Keywords: Arginine starvation, Epigenetic gene silencing, DNA leakage, cGAS-STING activation
 Global reach, higher impact
Global reach, higher impact