13.3
Impact Factor
Theranostics 2021; 11(12):5700-5712. doi:10.7150/thno.55163 This issue Cite
Research Paper
Externally triggered smart drug delivery system encapsulating idarubicin shows superior kinetics and enhances tumoral drug uptake and response
1. Laboratory Experimental Oncology, Department of Pathology, Erasmus MC, 3015GD Rotterdam, The Netherlands.
2. Department of Pediatrics, Medical University of South Carolina, Charleston, SC 29425, USA.
3. Erasmus MC Cancer Institute, Department of Radiation Oncology, 3015GD Rotterdam, The Netherlands.
4. Department of Pathology, Erasmus Optical Imaging Centre, Erasmus MC, 3000CA Rotterdam, The Netherlands.
Abstract
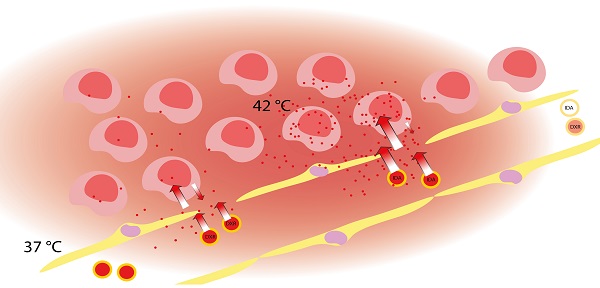
Rationale: Increasing the bioavailable drug level in a tumor is the key to enhance efficacy of chemotherapy. Thermosensitive smart drug delivery systems (SDDS) in combination with local hyperthermia facilitate high local drug levels, thus improving uptake in the tumor. However, inability to rapidly and efficiently absorb the locally released drug results in reduced efficacy, as well as undesired redistribution of the drug away from the tumor to the system.
Methods: Based on this paradigm we propose a novel approach in which we replaced doxorubicin (DXR), one of the classic drugs for nanocarrier-based delivery, with idarubicin (IDA), a hydrophobic anthracycline used solely in the free form for treatment hematologic cancers. We established a series of in vitro and in vivo experiments to in depth study the kinetics of SDDS-based delivery, drug release, intratumor biodistribution and subsequent cell uptake.
Results: We demonstrate that IDA is taken up over 10 times more rapidly by cancer cells than DXR in vitro. Similar trend is observed in in vivo online imaging and less drug redistribution is shown for IDA, together resulting in 4-times higher whole tumor drug uptake for IDA vs. DXR. Together his yielded an improved intratumoral drug distribution for IDA-SDDS, translating into superior tumor response compared to DXR-SDDS treatment at the same dose. Thus, IDA - a drug that is not used for treatment of solid cancers - shows superior therapeutic index and better outcome when administered in externally triggered SDDS.
Conclusions: We show that a shift in selection of chemotherapeutics is urgently needed, away from the classic drugs towards selection based on properties of a chemotherapeutic in context of the nanoparticle and delivery mode, to maximize the therapeutic efficacy.
Keywords: smart drug delivery system, triggered release, superior release kinetics, enhanced intratumoral uptake and distribution
 Global reach, higher impact
Global reach, higher impact