13.3
Impact Factor
Theranostics 2021; 11(5):2460-2474. doi:10.7150/thno.53169 This issue Cite
Research Paper
NEK9, a novel effector of IL-6/STAT3, regulates metastasis of gastric cancer by targeting ARHGEF2 phosphorylation
1. State Key Laboratory of Cancer Biology and National Clinical Research Center for Digestive Diseases, Xijing Hospital of Digestive Diseases, Fourth Military Medical University, Xi'an, China, 710032.
2. Department of Ultrasound Diagnostics, Tangdu Hospital, Fourth Military Medical University, Xi'an, China, 710032.
3. Department of Radiation Oncology, Xijing Hospital, Fourth Military Medical University, Xi'an, China, 710032.
#These authors contributed equally to this study.
Abstract
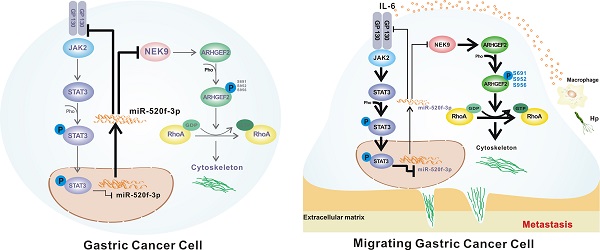
Rationale: Inflammatory stimuli from the tumor microenvironment play important roles in cancer progression. However, the mechanism of promotion of cancer metastasis by inflammation in gastric cancer (GC) is poorly understood.
Methods: The roles of NEK9 were validated via loss-of-function and gain-of-function experiments in vitro and in an animal model of metastasis. Cytoskeletal reorganization-associated molecules were detected by GST pull-down. The regulation of ARHGEF2 by NEK9 was investigated by phosphoproteomics analysis, immunoprecipitation (IP) and in vitro kinase assay. The transcriptional regulation of miR-520f-3p was studied using luciferase reporter and chromatin immunoprecipitation (ChIP). The expression of these proteins in GC tissues was examined by immunohistochemistry.
Results: NEK9 directly regulates cell motility and RhoA activation in GC. The phosphorylation of ARHGEF2 by NEK9 is the key step of this process. NEK9 is a direct target of miR-520f-3p, which is transcriptionally suppressed by IL-6-mediated activation of STAT3. A decrease in miR-520f-3p leads to the amplification of IL-6/STAT3 by targeting GP130. A simultaneous elevation of the levels of NEK9, GP130 and p-STAT3 was confirmed in the lymph nodes and distant metastases. An increase in NEK9, GP130 and STAT3 is associated with reduced overall survival of GC patients.
Conclusion: This study demonstrates that activation of STAT3 by IL-6 transcriptionally suppresses miR-520f-3p and diminishes the inhibitory effects of miR-520f-3p on NEK9 and GP130. An increase in GP130 enhances this signaling, and NEK9 directly influences cell motility and RhoA activation by targeting the phosphorylation of ARHGEF2. Targeting the IL-6-STAT3-NEK9 pathway may be a new strategy for GC treatment.
Keywords: Gastric cancer, Metastasis, NEK9, Inflammation, Phosphorylation
 Global reach, higher impact
Global reach, higher impact