13.3
Impact Factor
Theranostics 2021; 11(5):2334-2348. doi:10.7150/thno.49277 This issue Cite
Research Paper
Eradication of solid tumors by chemodynamic theranostics with H2O2-catalyzed hydroxyl radical burst
1. MOE Key Laboratory of Laser Life Science & Institute of Laser Life Science, College of Biophotonics, South China Normal University, Guangzhou, 510631, P.R. China
2. Guangdong Provincial Key Laboratory of Laser Life Science, College of Biophotonics, South China Normal University, Guangzhou, 510631, P.R. China
Abstract
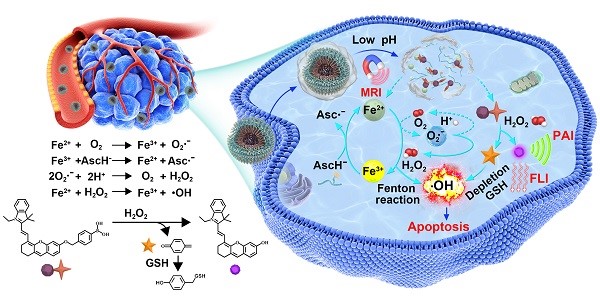
Activatable theranostics, integrating high diagnostic accuracy and significant therapeutic effect, holds great potential for personalized cancer treatments; however, their chemodynamic modality is rarely exploited. Herein, we report a new in situ activatable chemodynamic theranostics PAsc/Fe@Cy7QB to specifically recognize and eradicate cancer cells with H2O2-catalyzed hydroxyl radical (•OH) burst cascade.
Methods: The nanomicelles PAsc/Fe@Cy7QB were constructed by self-assembly of acid-responsive copolymers incorporating ascorbates and acid-sensitive Schiff base-Fe2+ complexes as well as H2O2-responsive adjuvant Cy7QB.
Results: Upon systematic delivery of PAsc/Fe@Cy7QB into cancer cells, the acidic microenvironment triggered disassembly of the nanomicelles. The released Fe2+ catalyzed the oxidation of ascorbate monoanion (AscH-) to efficiently produce H2O2. The released H2O2, together with the endogenous H2O2, could be converted into highly active •OH via the Fenton reaction, resulting in enhanced Fe-mediated T1 magnetic resonance imaging (MRI). The synchronously released Cy7QB was activated by H2O2 to produce a glutathione (GSH)-scavenger quinone methide to boost the •OH yield and recover the Cy7 dye for fluorescence and photoacoustic imaging.
Conclusion: The biodegradable PAsc/Fe@Cy7QB designed for tumor-selective multimodal imaging and high therapeutic effect provides an exemplary paradigm for precise chemodynamic theranostic.
Keywords: chemodynamic theranostics, activatable, reactive oxygen species, Fenton reaction, multimodal imaging
 Global reach, higher impact
Global reach, higher impact