13.3
Impact Factor
Theranostics 2021; 11(1):379-396. doi:10.7150/thno.50028 This issue Cite
Research Paper
Tumor cell-activated “Sustainable ROS Generator” with homogeneous intratumoral distribution property for improved anti-tumor therapy
1. School of Pharmaceutical Sciences, Zhengzhou University, Zhengzhou 450001, China.
2. Key Laboratory of Targeting Therapy and Diagnosis for Critical Diseases, Henan Province, China, Zhengzhou University, Zhengzhou 450001, China.
3. Collaborative Innovation Center of New Drug Research and Safety Evaluation, Henan Province, Zhengzhou, China, Zhengzhou University, Zhengzhou 450001, China.
*These authors contributed equally to this work.
Abstract
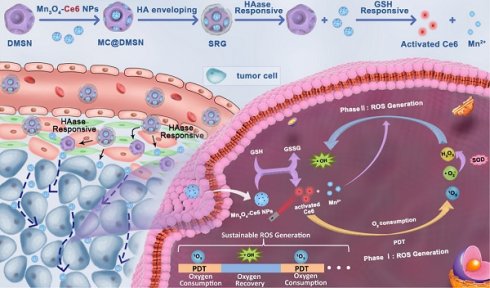
Photodynamic therapy (PDT) holds a number of advantages for tumor therapy. However, its therapeutic efficiency is limited by non-sustainable reactive oxygen species (ROS) generation and heterogeneous distribution of photosensitizer (PS) in tumor. Herein, a “Sustainable ROS Generator” (SRG) is developed for efficient antitumor therapy.
Methods: SRG was prepared by encapsulating small-sized Mn3O4-Ce6 nanoparticles (MC) into dendritic mesoporous silica nanoparticles (DMSNs) and then enveloped with hyaluronic acid (HA). Due to the high concentration of HAase in tumor tissue, the small-sized MC could be released from DMSNs and homogeneously distributed in whole tumor. Then, the released MC would be uptaken by tumor cells and degraded by high levels of intracellular glutathione (GSH), disrupting intracellular redox homeostasis. More importantly, the released Ce6 could efficiently generate singlet oxygen (1O2) under laser irradiation until the tissue oxygen was exhausted, and the manganese ion (Mn2+) generated by degraded MC would then convert the low toxic by-product (H2O2) of PDT to the most harmful ROS (·OH) for sustainable and recyclable ROS generation.
Results: MC could be homogeneously distributed in whole tumor and significantly reduced the level of intracellular GSH. At 2 h after PDT, obvious intracellular ROS production was still observed. Moreover, during oxygen recovery in tumor tissue, ·OH could be continuously produced, and the nanosystem could induce 82% of cell death comparing with 30% of cell death induced by free Ce6. For in vivo PDT, SRG achieved a complete inhibition on tumor growth.
Conclusion: Based on these findings, we conclude that the designed SRG could induce sustainable ROS generation, homogeneous intratumoral distribution and intracellular redox homeostasis disruption, presenting an efficient strategy for enhanced ROS-mediated anti-tumor therapy.
Keywords: sustainable ROS generation, homogeneous tumor distribution, switched on, GSH depletion
 Global reach, higher impact
Global reach, higher impact