13.3
Impact Factor
Theranostics 2020; 10(19):8744-8756. doi:10.7150/thno.45845 This issue Cite
Research Paper
Erythropoietin receptor in B cells plays a role in bone remodeling in mice
1. Department of Cell and Developmental Biology, Sackler Faculty of Medicine, Tel Aviv University, Israel.
2. Department of Anatomy and Anthropology, Sackler Faculty of Medicine, Tel Aviv University, Israel.
3. Department of Medicine A, Tel Aviv Sourasky Medical Center, Sackler Faculty of Medicine, Tel Aviv University, Israel.
4. Department of Medicine III, Dresden University Medical Center, Germany.
5. Institute for Clinical Chemistry and Laboratory Medicine, Technische Universität Dresden, Dresden, Germany.
*Equal contribution
Abstract
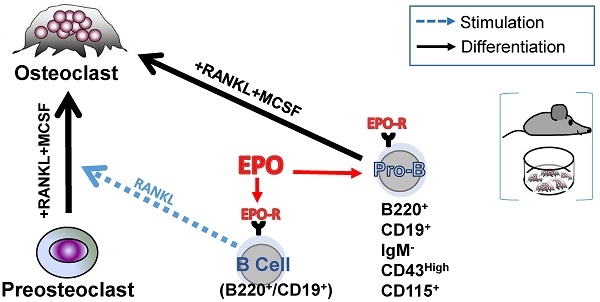
Erythropoietin (EPO) is a key regulator of erythropoiesis. However, EPO receptors (EPO-Rs) are also expressed on non-erythroid cell types, including myeloid and bone cells. Immune cells also participate in bone homeostasis. B cells produce receptor activator of nuclear factor kappa-Β ligand (RANKL) and osteoprotegerin (OPG), two pivotal regulators of bone metabolism. Here we explored the ability of B cells to transdifferentiate into functional osteoclasts and examined the role of EPO in this process in a murine model.
Methods: We have combined specifically-designed experimental mouse models and in vitro based osteoclastogenesis assays, as well as PCR analysis of gene expression.
Results: (i) EPO treatment in vivo increased RANKL expression in bone marrow (BM) B cells, suggesting a paracrine effect on osteoclastogenesis; (ii) B cell-derived osteoclastogenesis occured in vivo and in vitro, as demonstrated by B cell lineage tracing in murine models; (iii) B-cell-derived osteoclastogenesis in vitro was restricted to Pro-B cells expressing CD115/CSF1-R and is enhanced by EPO; (iv) EPO treatment increased the number of B-cell-derived preosteoclasts (β3+CD115+), suggesting a physiological rationale for B cell derived osteoclastogenesis; (v) finally, mice with conditional EPO-R knockdown in the B cell lineage (cKD) displayed a higher cortical and trabecular bone mass. Moreover, cKD displayed attenuated EPO-driven trabecular bone loss, an effect that was observed despite the fact that cKD mice attained higher hemoglobin levels following EPO treatment.
Conclusions: Our work highlights B cells as an important extra-erythropoietic target of EPO-EPO-R signaling and suggests their involvement in the regulation of bone homeostasis and possibly in EPO-stimulated erythropoietic response. Importantly, we present here for the first time, histological evidence for B cell-derived osteoclastogenesis in vivo.
Keywords: bone marrow, Pro-B cells, lymphocytes, osteoclastogenesis, transdifferentiation, erythropoietin, cFMS/CD115/CSF1R
 Global reach, higher impact
Global reach, higher impact