13.3
Impact Factor
Theranostics 2020; 10(19):8691-8704. doi:10.7150/thno.45392 This issue Cite
Research Paper
Self-accelerating H2O2-responsive Plasmonic Nanovesicles for Synergistic Chemo/starving therapy of Tumors
1. State Key Laboratory of Bioreactor Engineering, Shanghai Key Laboratory of Chemical Biology, School of Pharmacy, East China University of Science and Technology, Shanghai 200237, China.
2. State Key Laboratory of Molecular Engineering of Polymers, Department of Macromolecular Science, Fudan University, Shanghai 200438, China.
3. Department of Pharmacy and Pharmacology, University of the Witwatersrand, Parktown 2193 Johannesburg, South Africa.
4. Department of Emergency Medicine, the General Hospital of Northern Theater Command, Laboratory of Rescue Center of Severe Trauma PLA, Shenyang l10016, China.
Abstract
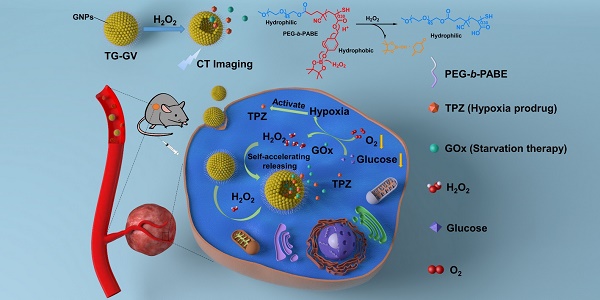
Rationale: Nanoscale vehicles responsive to abnormal variation in tumor environment are promising for use in targeted delivery of therapeutic drugs specifically to tumor sites. Herein, we report the design and fabrication of self-accelerating H2O2-responsive plasmonic gold nanovesicles (GVs) encapsulated with tirapazamine (TPZ) and glucose oxidase (GOx) for synergistic chemo/starving therapy of cancers.
Methods: Gold nanoparticles were modified with H2O2-responsive amphiphilic block copolymer PEG45-b-PABE330 by ligand exchange. The TPZ and GOx loaded GVs (TG-GVs) were prepared through the self-assembly of PEG45-b-PABE330 -grafted nanoparticles together with TPZ and GOx by solvent displacement method.
Results: In response to H2O2 in tumor, the TG-GVs dissociate to release the payloads that are, otherwise, retained inside the vesicles for days without noticeable leakage. The released GOx enzymes catalyze the oxidation of glucose by oxygen in the tumor tissue to enhance the degree of hypoxia that subsequently triggers the reduction of hypoxia-activated pro-drug TPZ into highly toxic free radicals. The H2O2 generated in the GOx-catalyzed reaction also accelerate the dissociation of vesicles and hence the release rate of the cargoes in tumors. The drug-loaded GVs exhibit superior tumor inhibition efficacy in 4T1 tumor-bearing mice owing to the synergistic effect of chemo/starvation therapy, in addition to their use as contrast agents for computed tomography imaging of tumors.
Conclusion: This nanoplatform may find application in managing tumors deeply trapped in viscera or other important tissues that are not compatible with external stimulus (e.g. light).
Keywords: hydrogen peroxide, gold vesicles, cancer therapy, controlled release, cancer imaging, self-accelerating
 Global reach, higher impact
Global reach, higher impact