Impact Factor
Theranostics 2020; 10(19):8573-8590. doi:10.7150/thno.44321 This issue Cite
Research Paper
HSPA12A unstabilizes CD147 to inhibit lactate export and migration in human renal cell carcinoma
1. Department of Anesthesiology, Jiangsu Provincial Key Laboratory of Geriatrics, the First Affiliated Hospital with Nanjing Medical University, Nanjing 210029, China;
2. Department of Geriatrics, First Affiliated Hospital with Nanjing Medical University, Nanjing 210029, China;
3. Key Laboratory of Targeted Intervention of Cardiovascular Disease, Collaborative Innovation Center for Cardiovascular Disease Translational Medicine, Nanjing Medical University, Nanjing 210029, China;
4. Department of Surgery, East Tennessee State University, Johnson City, TN 37614.
Abstract
Background: Metastasis accounts for 90% of cancer-associated mortality in patients with renal cell carcinoma (RCC). However, the clinical management of RCC metastasis is challenging. Lactate export is known to play an important role in cancer cell migration. This study investigated the role of heat shock protein A12A (HSPA12A) in RCC migration.
Methods: HSPA12A expression was examined in 82 pairs of matched RCC tumors and corresponding normal kidney tissues from patients by immunoblotting and immunofluorescence analyses. The proliferation of RCC cells was analyzed using MTT and EdU incorporation assays. The migration of RCC cells was evaluated by wound healing and Transwell migration assays. Extracellular acidification was examined using Seahorse technology. Protein stability was determined following treatment with protein synthesis inhibitor cycloheximide and proteasome inhibitor MG132. Mass spectrometry, immunoprecipitation, and immunoblotting were employed to examine protein-protein interactions.
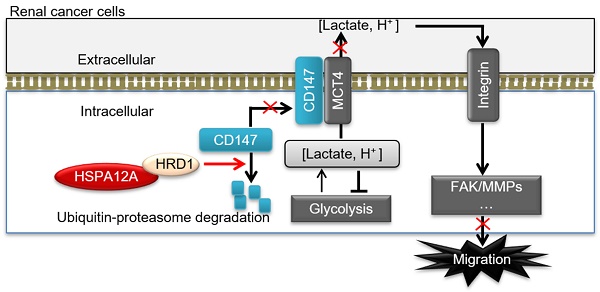
Results: RCC tumors from patients showed downregulation of HSPA12A, which was associated with advanced tumor node metastasis stage. Intriguingly, overexpression of HSPA12A in RCC cells inhibited migration, whereas HSPA12A knockdown had the opposite effect. Lactate export, glycolysis rate, and CD147 protein abundance were also inhibited by HSPA12A overexpression but promoted by HSPA12A knockdown. An interaction of HSPA12A with HRD1 ubiquitin E3 ligase was detected in RCC cells. Further studies demonstrated that CD147 ubiquitination and proteasomal degradation were promoted by HSPA12A overexpression whereas inhibited by HSPA12A knockdown. Notably, the HSPA12A overexpression-induced inhibition of lactate export and migration were abolished by CD147 overexpression.
Conclusion: Human RCC shows downregulation of HSPA12A. Overexpression of HSPA12A in RCC cells unstabilizes CD147 through increasing its ubiquitin-proteasome degradation, thereby inhibits lactate export and glycolysis, and ultimately suppresses RCC cell migration. Our results demonstrate that overexpression of HSPA12A might represent a viable strategy for managing RCC metastasis.
Keywords: renal cell carcinoma, heat shock protein A12A (HSPA12A), migration, cluster of differentiation 147 (CD147), lactate
 Global reach, higher impact
Global reach, higher impact