13.3
Impact Factor
Theranostics 2020; 10(16):7369-7383. doi:10.7150/thno.44893 This issue Cite
Research Paper
Decreased Neuronal Excitability in Medial Prefrontal Cortex during Morphine Withdrawal is associated with enhanced SK channel activity and upregulation of small GTPase Rac1
1. Department of Neurosurgery, Tangdu Hospital, The Fourth Military Medical University, Xi'an, Shaanxi, 710038, China.
2. Laboratory of Neuroimaging, National Institute on Alcohol Abuse and Alcoholism, Bethesda, MD, 20892-1013, USA.
3. National Institute on Drug Abuse, National Institutes of Health, Bethesda, MD, 20892, USA.
4. School of Medicine, Medical Sciences and Nutrition, Institute of Medical Sciences, University of Aberdeen, Aberdeen, United Kingdom.
5. National Clinical Research Center for Mental Disorders, Changsha, 410013, China.
6. Department of Neurosurgery, The First Affiliated Hospital of Xi'an Jiaotong University, Xi'an, 710061, China.
7. Department of Neurosurgery, Xinjiang Production and Construction Corps Hospital, Xinjiang, 830002, China.
*These authors contributed equally to this study.
Abstract
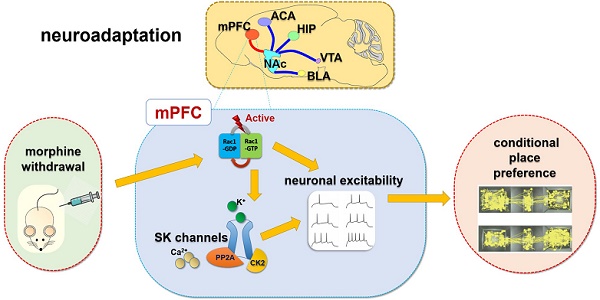
Rationale: Neuroadaptations in the medial prefrontal cortex (mPFC) and Nucleus Accumbens (NAc) play a role in the disruption of control-reward circuits in opioid addiction. Small Conductance Calcium-Activated Potassium (SK) channels in the mPFC have been implicated in neuronal excitability changes during morphine withdrawal. However, the mechanism that modulates SK channels during withdrawal is still unknown.
Methods: Rats were exposed for one week to daily morphine injections (10 mg·kg-1 s.c.) followed by conditional place preference (CPP) assessment. One week after withdrawal, electrophysiological, morphological and molecular biological methods were applied to investigate the effects of morphine on SK channels in mPFC, including infralimbic (IL), prelimbic (PrL) cortices and NAc (core and shell). We verified the hypothesis that Rac1, a member of Rho family of small GTPases, implicated in SK channel regulation, modulate SK channel neuroadaptations during opiate withdrawal.
Results: One week after morphine withdrawal, the neuronal excitability of layer 5 pyramidal neurons in IL was decreased, but not in PrL. Whereas, the excitability was increased in NAc-shell, but not in NAc-core. In mPFC, the expression of the SK3 subunit was enhanced after one-week of withdrawal compared to controls. In the IL, Rac1 signaling was increased during withdrawal, and the Rac1 inhibitor NSC23766 disrupted SK current, which increased neuronal firing. Suppression of Rac1 inhibited morphine-induced CPP and expression of SK channels in IL.
Conclusions: These findings highlight the potential value of SK channels and the upstream molecule Rac1, which may throw light on the therapeutic mechanism of neuromodulation treatment for opioid dependence.
Keywords: Morphine, Nucleus Accumbens (NAc), medial prefrontal cortex (mPFC), Small Conductance Calcium-Activated Potassium Channels (SK channels), Rac1
 Global reach, higher impact
Global reach, higher impact