13.3
Impact Factor
Theranostics 2020; 10(14):6361-6371. doi:10.7150/thno.46468 This issue Cite
Research Paper
Comparative study of preclinical mouse models of high-grade glioma for nanomedicine research: the importance of reproducing blood-brain barrier heterogeneity
1. Australian Institute for Bioengineering and Nanotechnology, The University of Queensland, Brisbane, Australia
2. ARC Centre of Excellence in Convergent Bio-Nano Science and Technology, The University of Queensland, Brisbane, Australia
3. Australian e-Health Research Centre, Commonwealth Scientific and Industrial Research Organization, Royal Brisbane and Women's Hospital, Brisbane, Australia
4. Institute for Molecular Bioscience, The University of Queensland, Brisbane, Australia
5. Department of Cell and Molecular Biology, QIMR Berghofer Medical Research Institute, Brisbane, Australia
6. School of Biomedical Sciences, Faculty of Health, Queensland University of Technology, Brisbane, Australia
7. School of Biomedical Sciences, The University of Queensland, Brisbane, Australia
✉ Corresponding author: Simon Puttick, MSci, PhD, Australian e-Health Research Centre, Commonwealth Scientific and Industrial Research Organization, Level 5 UQ Health Sciences Building, Royal Brisbane and Women's Hospital, Herston QLD 4029, Australia. Telephone: +61 (0)732533638 E-mail: simon.puttick@csiro.au.
Abstract
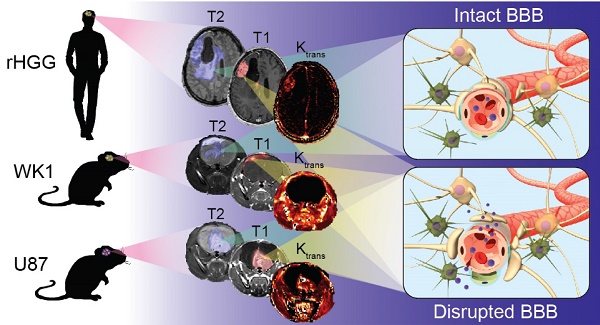
The clinical translation of new nanoparticle-based therapies for high-grade glioma (HGG) remains extremely poor. This has partly been due to the lack of suitable preclinical mouse models capable of replicating the complex characteristics of recurrent HGG (rHGG), namely the heterogeneous structural and functional characteristics of the blood-brain barrier (BBB). The goal of this study is to compare the characteristics of the tumor BBB of rHGG with two different mouse models of HGG, the ubiquitously used U87 cell line xenograft model and a patient-derived cell line WK1 xenograft model, in order to assess their suitability for nanomedicine research.
Method: Structural MRI was used to assess the extent of BBB opening in mouse models with a fully developed tumor, and dynamic contrast enhanced MRI was used to obtain values of BBB permeability in contrast enhancing tumor. H&E and immunofluorescence staining were used to validate results obtained from the in vivo imaging studies.
Results: The extent of BBB disruption and permeability in the contrast enhancing tumor was significantly higher in the U87 model than in rHGG. These values in the WK1 model are similar to those of rHGG. The U87 model is not infiltrative, has an entirely abnormal and leaky vasculature and it is not of glial origin. The WK1 model infiltrates into the non-neoplastic brain parenchyma, it has both regions with intact BBB and regions with leaky BBB and remains of glial origin.
Conclusion: The WK1 mouse model more accurately reproduces the extent of BBB disruption, the level of BBB permeability and the histopathological characteristics found in rHGG patients than the U87 mouse model, and is therefore a more clinically relevant model for preclinical evaluations of emerging nanoparticle-based therapies for HGG.
Keywords: High-grade glioma, blood-brain barrier, vascular permeability, nanoparticle-based therapies, preclinical mouse model
 Global reach, higher impact
Global reach, higher impact