Impact Factor
Theranostics 2020; 10(11):4903-4928. doi:10.7150/thno.42480 This issue Cite
Review
Esophageal, gastric and colorectal cancers: Looking beyond classical serological biomarkers towards glycoproteomics-assisted precision oncology
1. Experimental Pathology and Therapeutics Group, Portuguese Institute of Oncology, 4200-162 Porto, Portugal;
2. Institute of Biomedical Sciences Abel Salazar (ICBAS), University of Porto, 4050-013 Porto, Portugal;
3. Institute for Research and Innovation in Health (i3s) , University of Porto, 4200-135 Porto, Portugal;
4. Institute for Biomedical Engineering (INEB), Porto, Portugal, 4200-135 Porto, Portugal;
5. Digestive Cancer Research Group, 1495-161 Algés, Portugal;
6. Porto Comprehensive Cancer Centre (P.ccc), 4200-162 Porto, Portugal;
7. QOPNA/LAQV, Departamento de Química da Universidade de Aveiro, Aveiro, Portugal;
8. Department of Surgical Oncology, Portuguese Institute of Oncology of Porto, 4200-162 Porto, Portugal;
9. Institute of Molecular Pathology and Immunology of the University of Porto (IPATIMUP), Porto, Portugal;
10. Faculty of Medicine, University of Porto, Porto, Portugal.
*Equal contribution
Abstract
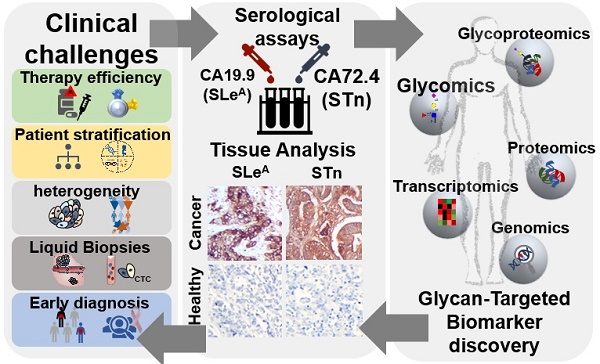
Esophageal (OC), gastric (GC) and colorectal (CRC) cancers are amongst the digestive track tumors with higher incidence and mortality due to significant molecular heterogeneity. This constitutes a major challenge for patients' management at different levels, including non-invasive detection of the disease, prognostication, therapy selection, patient's follow-up and the introduction of improved and safer therapeutics. Nevertheless, important milestones have been accomplished pursuing the goal of molecular-based precision oncology. Over the past five years, high-throughput technologies have been used to interrogate tumors of distinct clinicopathological natures, generating large-scale biological datasets (e.g. genomics, transcriptomics, and proteomics). As a result, GC and CRC molecular subtypes have been established to assist patient stratification in the clinical settings. However, such molecular panels still require refinement and are yet to provide targetable biomarkers. In parallel, outstanding advances have been made regarding targeted therapeutics and immunotherapy, paving the way for improved patient care; nevertheless, important milestones towards treatment personalization and reduced off-target effects are also to be accomplished. Exploiting the cancer glycoproteome for unique molecular fingerprints generated by dramatic alterations in protein glycosylation may provide the necessary molecular rationale towards this end. Therefore, this review presents functional and clinical evidences supporting a reinvestigation of classical serological glycan biomarkers such as sialyl-Tn (STn) and sialyl-Lewis A (SLeA) antigens from a tumor glycoproteomics perspective. We anticipate that these glycobiomarkers that have so far been employed in non-invasive cancer prognostication may hold unexplored value for patients' management in precision oncology settings.
Keywords: glycobiomarkers, glycoproteomics, glycomics, glycosylation, digestive tract cancer, precision oncology
 Global reach, higher impact
Global reach, higher impact