13.3
Impact Factor
Theranostics 2020; 10(11):4779-4794. doi:10.7150/thno.43736 This issue Cite
Research Paper
Enhanced osseointegration of three-dimensional supramolecular bioactive interface through osteoporotic microenvironment regulation
1. Orthopaedic Medical Center, The Second Hospital of Jilin University, Changchun 130041, P. R. China
2. State Key Lab of Supramolecular Structure and Materials, College of Chemistry, Jilin University, Changchun 130012, P. R. China
3. Orthopaedic Research Institute of Jilin Province, Changchun 130041, P. R. China
4. Department of Plastic and Reconstruct Surgery, The First Bethune Hospital of Jilin University, Changchun 130021, P. R. China
5. Department of Pain, Renji Hospital, South Campus, Shanghai Jiaotong University, Shanghai 201112, P. R. China
*Haotian Bai and Yue Zhao contributed equally to this work.
Abstract
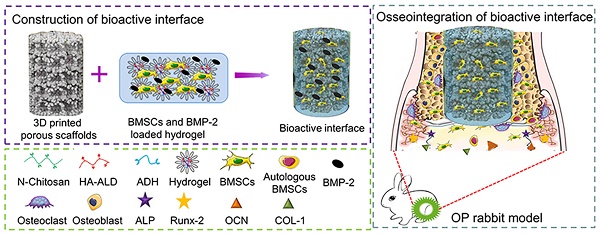
Purpose: Osteoporosis is more likely to cause serious complications after joint replacement, mainly due to physiological defects of endogenous osteogenic cells and the pathological osteoclast activity. It is a feasible solution to design a prosthetic surface interface that specifically addresses this troublesome situation.
Methods: A novel “three-dimensional (3D) inorganic-organic supramolecular bioactive interface” was constructed consisting of stiff 3D printing porous metal scaffold and soft multifunctional, self-healable, injectable, and biodegradable supramolecular polysaccharide hydrogel. Apart from mimicking the bone extracellular matrix, the bioactive interface could also encapsulate bioactive substances, namely bone marrow mesenchymal stem cells (BMSCs) and bone morphogenetic protein-2 (BMP-2). A series of in vitro characterizations, such as topography and mechanical characterization, in vitro release of BMP-2, biocompatibility analysis, and osteogenic induction of BMSCs were carried out. After that, the in vivo osseointegration effect of the bioactive interface was investigated in detail using an osteoporotic model.
Results: The administration of injectable supramolecular hydrogel into the inner pores of 3D printing porous metal scaffold could obviously change the morphology of BMSCs and facilitate its cell proliferation. Meanwhile, BMP-2 was capable of being sustained released from supramolecular hydrogel, and subsequently induced osteogenic differentiation of BMSCs and promoted the integration of the metal microspores-bone interface in vitro and in vivo. Moreover, the osteoporosis condition of bone around the bioactive interface was significantly ameliorated.
Conclusion: This study demonstrates that the 3D inorganic-organic supramolecular bioactive interface can serve as a novel artificial prosthesis interface for various osteogenesis-deficient patients, such as osteoporosis and rheumatoid arthritis.
Keywords: bioactive interface, bone morphogenetic protein 2, osteoporotic microenvironment, osseointegration, supramolecular hydrogel
 Global reach, higher impact
Global reach, higher impact