13.3
Impact Factor
Theranostics 2020; 10(11):4737-4748. doi:10.7150/thno.42565 This issue Cite
Review
Size profile of cell-free DNA: A beacon guiding the practice and innovation of clinical testing
1. National Center for Clinical Laboratories, Beijing Hospital, National Center of Gerontology; Institute of Geriatric Medicine, Chinese Academy of Medical Sciences, Beijing, People's Republic of China
2. Graduate School, Peking Union Medical College, Chinese Academy of Medical Sciences, Beijing 100730, People's Republic of China
3. Peking University Fifth School of Clinical Medicine, Beijing Hospital, Beijing, People's Republic of China
4. Beijing Engineering Research Center of Laboratory Medicine, Beijing, People's Republic of China
* Jiping Shi and Runling Zhang contributed equally to the work.
Abstract
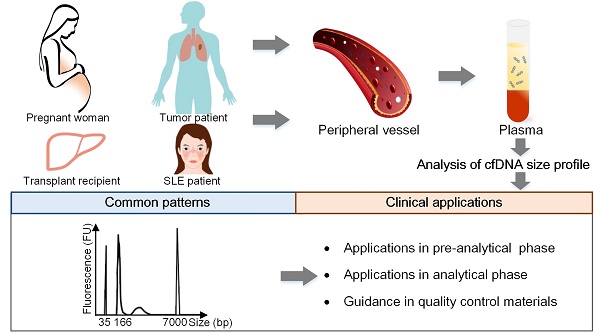
Cell-free DNA (cfDNA) has pioneered the development of noninvasive prenatal testing and liquid biopsy, its emerging applications include organ transplantation, autoimmune diseases, and many other disorders; size profile of cfDNA is a crucial biological property and is essential for its clinical applications. Therefore, a thorough mastery of the characteristic and potential applications of cfDNA size profile is needed.
Methods: Based on the recent researches, we summarized the size profile of cfDNA in pregnant women, tumor patients, transplant recipients and systemic lupus erythematosus (SLE) patients to explore the common features. We also concluded the applications of size profile in pre-analytical phases, analytical phases for novel assays, and preparation of quality control materials (QCMs).
Results: The size profile of cfDNA shared common features in different populations, and was distributed as a “ladder” pattern with a dominant peak at ~166 bp. However, cfDNA entailed slightly discrepant characteristics due to specific tissues of origin. The dominant peaks of fetal and maternal cfDNA fragments in pregnant women were at 143 bp and 166 bp, respectively. The plasma cfDNA in tumor patients, transplant recipients, and SLE patients had a peak of around 166 bp. In pre-analytical phases, size profile served as a vital indicator to judge the eligibility of specimens, thus ensuring the successful implementation of assays. More importantly, the size profile had the potential to enrich short fragments, calculate fetal fraction, detect fetal abnormalities, predict tumor progress in analytical phase and to guide the preparation of QCMs.
Conclusions: Our finding summarized the characteristics and potential applications of cfDNA size profile, providing clinical researchers with novel assays by the extensive application of cfDNA.
Keywords: cell-free DNA, size profile, mechanism, clinical application, quality control
 Global reach, higher impact
Global reach, higher impact