Impact Factor
Theranostics 2020; 10(8):3594-3611. doi:10.7150/thno.40318 This issue Cite
Research Paper
Mucoadhesive-to-penetrating controllable peptosomes-in-microspheres co-loaded with anti-miR-31 oligonucleotide and Curcumin for targeted colorectal cancer therapy
1. State Key Laboratories for Agrobiotechnology and Beijing Advanced Innovation Center for Food Nutrition and Human Health, College of Biological Sciences, China Agricultural University, Beijing, 100193, China.
2. Guangdong Provincial Key Laboratory of Regional Immunity and School of Medicine, Shenzhen University, Shenzhen, 518055, China.
3. Beijing Advanced Innovation Center for Food Nutrition and Human Health, Key Laboratory of Functional Dairy, College of Food Science and Nutritional Engineering, China Agricultural University, Beijing, 100083, China.
4. College of Veterinary Medicine, China Agricultural University, Beijing, 100193, China.
5. CAS Key Laboratory of Bio-medical Diagnostics, Suzhou Institute of Biomedical Engineering and Technology, Chinese Academy of Sciences, Suzhou, Jiangsu, 215163, China.
6. Institute of Reproductive and Developmental Biology, Faculty of Medicine, Imperial College London, W12 0NN, UK.
7. Department of Developmental and Cell Biology, Sue and Bill Gross Stem Cell Research center, Center for Complex Biological Systems, University of California, Irvine, Irvine, CA 92697, USA.
8. Division of Gastroenterology, Union Hospital, Tongji Medical College, Huazhong University of Technology and Science, Wuhan, 430022, China.
9. Department of Gastroenterology, Xijing Hospital, The Fourth Military Medical University, Xi'an, 710032, China.
10. Department of Gastroenterology, The Shanghai Tenth People's Hospital, Tongji University, Shanghai, 200072, China.
11. State Key Laboratory of Molecular Oncology, National Cancer Center/Cancer Hospital, Chinese Academy of Medical Sciences, Beijing, 100021, China.
12. Department of Microbiology and Immunology, University of Texas Medical Branch, Galveston, TX 78701, USA.
#These authors contributed equally to this work.
Abstract
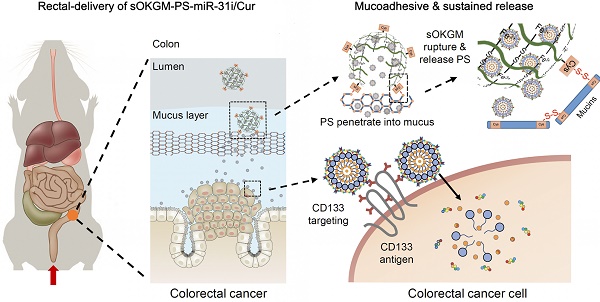
Background: Accumulating evidences indicate that nanomedicines greatly decrease the side effects and enhance the efficacy of colorectal cancer (CRC) treatment. In particular, the use of rectal delivery of nanomedicines, with advantages such as fast therapeutic effects and a diminishing hepatic first-pass effect, is currently emerging.
Method: We established a CRC targeted delivery system, in which α-lactalbumin peptosomes (PSs) co-loaded with a microRNA (miR)-31 inhibitor (miR-31i) and curcumin (Cur) were encapsuslated in thiolated TEMPO oxidized Konjac glucomannan (sOKGM) microspheres, referred as sOKGM-PS-miR-31i/Cur. The CRC targeting capability, drug release profiles, mucoadhesive-to-penetrating properties and therapeutic efficacy of sOKGM-PS-miR-31i/Cur delivery system were evaluated in colorectal cancer cells and azoxymethane-dextran sodium (AOM-DSS) induced tumor models.
Results: sOKGM-PS-miR-31i/Cur delivery system were stable in the harsh gastrointestinal environment after rectal or oral administration; and were also mucoadhesive due to disulfide bond interactions with the colonic mucus layer, resulting in an enhanced drug retention and local bioavailability in the colon. Concomitantly, the released PS-miR-31i/Cur PSs from the microsphere was mucus-penetrating, efficiently passing through the colonic mucus layer, and allowed Cur and miR-31i specifically target to colon tumor cells with the guide of CD133 targeting peptides. Consequently, rectal delivery of sOKGM-PS-miR-31i/Cur microspheres suppressed tumor growth in an azoxymethane-dextran sodium sulfate (AOM-DSS)-induced tumor model.
Conclusion: sOKGM-PS-miR-31i/Cur microspheres are effective rectal delivery system with combined advantages of mucoadhesive and mucus-penetrating properties, representing a potent and viable therapeutic approach for CRC.
Keywords: Peptosomes-in-microsphere, Mucoadhesive-to-penetrating, Rectal delivery, Oral delivery, Anti-microRNA oligonucleotide, Anti-colorectal cancer therapy.
 Global reach, higher impact
Global reach, higher impact