13.3
Impact Factor
Theranostics 2020; 10(7):2930-2942. doi:10.7150/thno.38702 This issue Cite
Research Paper
Integrin αvβ6-specific therapy for pancreatic cancer developed from foot-and-mouth-disease virus
1. Barts Cancer Institute, Queen Mary University of London, John Vane Science Centre, Charterhouse Square, London EC1M 6BQ, UK.
2. Cancer Research UK Centre for Epidemiology, Mathematics and Statistics, Wolfson Institute of Preventative Medicine, Queen Mary University of London, Charterhouse Square, London EC1M 6BQ, UK.
3. Spirogen, QMB Innovation Centre, 42 New Road, London E1 2AX, UK.
4. ADC Therapeutics (UK) Ltd, QMB Innovation Centre, 42 New Road, London E1 2AX, UK.
5. Cancer Research UK Drug-DNA Interactions Research Group, University College London Cancer Institute, 72 Huntley Street, London WC1E 6BT, U.K.
Abstract
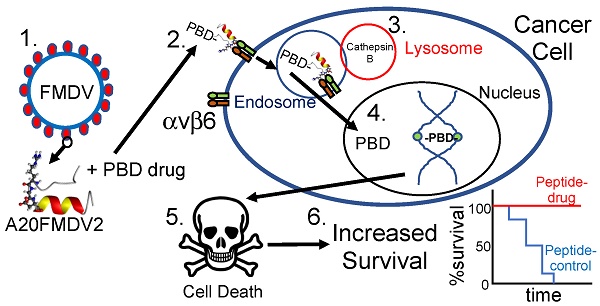
Goals of investigation: The 5-year survival rate for pancreatic ductal adenocarcinoma (PDAC) has remained at <5% for decades because no effective therapies have been identified. Integrin αvβ6 is overexpressed in most PDAC and represents a promising therapeutic target. Thus, we attempted to develop an αvβ6-specific peptide-drug conjugate (PDC) for therapy of PDAC.
Methodology: We conjugated the DNA-binding pyrrolobenzodiazepine (PBD)-based payload SG3249 (tesirine) to an αvβ6-specific 20mer peptide from the VP1 coat protein of foot-and-mouth-disease virus (FMDV) (forming conjugate SG3299) or to a non-targeting peptide (forming conjugate SG3511). PDCs were tested for specificity and toxicity on αvβ6-negative versus-positive PDAC cells, patient-derived cell lines from tumor xenografts, and on two different in vivo models of PDAC. Immunohistochemical analyses were performed to establish therapeutic mechanism.
Results: The αvβ6-targeted PDC SG3299 was significantly more toxic (up to 78-fold) for αvβ6-expressing versus αvβ6-negative PDAC cell lines in vitro, and achieved significantly higher toxicity at equal dose than the non-targeted PDC SG3511 (up to 15-fold better). Moreover, SG3299 eliminated established (100mm3) Capan-1 PDAC human xenografts, extending the lifespan of mice significantly (P=0.005). Immunohistochemistry revealed SG3299 induced DNA damage and apoptosis (increased γH2AX and cleaved caspase 3, respectively) associated with significant reductions in proliferation (Ki67), β6 expression and PDAC tumour growth.
Conclusions: The FMDV-peptide drug conjugate SG3299 showed αvβ6-selectivity in vitro and in vivo and can specifically eliminate αvβ6-positive cancers, providing a promising new molecular- specific therapy for pancreatic cancer.
Keywords: integrin, αvβ6, PDAC, peptide-drug conjugate
 Global reach, higher impact
Global reach, higher impact