13.3
Impact Factor
Theranostics 2020; 10(4):1833-1848. doi:10.7150/thno.39814 This issue Cite
Research Paper
UCH-L1-mediated Down-regulation of Estrogen Receptor α Contributes to Insensitivity to Endocrine Therapy for Breast Cancer
1. Xiangya School of Pharmaceutical Sciences, Central South University, Changsha, 410013, China
2. Department of Pharmacy, the Second Xiangya Hospotal, Central South University, Changsha, 410011, China
3. Department of Pathology, Xiangya hospital and Department of Pathology, School of Basic Medicine, Central South University, Changsha, 410078, China
4. Cancer Research Institute, School of Basic Medicine, and Key Laboratory of Carcinogenesis and Cancer Invasion, Ministry of Education, Central South University, Changsha, 410078, China
5. Department of Breast and Thyroid, The Third Xiangya Hospital, Central South University, Changsha, 410078, China
6. Department of Dermatology, Hunan Engineering Research Center of Skin Health and Disease, Hunan Key Laboratory of Skin Cancer and Psoriasis, Xiangya Hospital, Central South University, Changsha, Hunan 410008, China
7. Department of Cancer Biology and Toxicology, College of Medicine, Markey Cancer Center, University of Kentucky, Lexington, KY 40536, USA
8. Department of Pharmacology, College of Pharmaceutical Sciences, Soochow University, Suzhou, China, 215000, China
9. Key Laboratory of Animal Models and Human Disease Mechanisms of Chinese Academy of Sciences and Yunnan Province, Kunming Institute of Zoology, Chinese Academy of Sciences, Collaborative Innovation Center for Cancer Medicine, Kunming, 650223, China
10. Department of Internal Medicine, University of Iowa Carver College of Medicine, Iowa City, IA, 52242,USA.
Xi-Sha Chen and Kuan-Song Wang contributed equally to this work.
Abstract
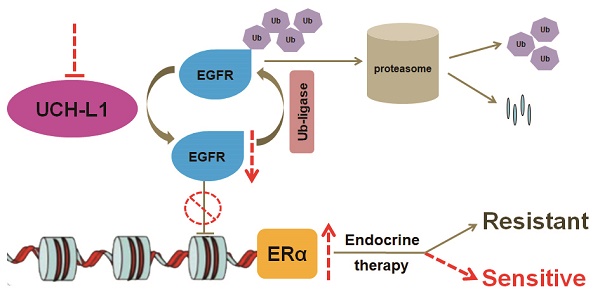
Purpose: To determine the role of UCH-L1 in regulating ERα expression, and to evaluate whether therapeutic targeting of UCH-L1 can enhance the efficacy of anti-estrogen therapy against breast cancer with loss or reduction of ERα.
Methods: Expressions of UCH-L1 and ERα were examined in breast cancer cells and patient specimens. The associations between UCH-L1 and ERα, therapeutic response and prognosis in breast cancer patients were analyzed using multiple databases. The molecular pathways by which UCH-L1 regulates ERα were analyzed using immunoblotting, qRT-PCR, immunoprecipitation, ubiquitination, luciferase and ChIP assays. The effects of UCH-L1 inhibition on the efficacy of tamoxifen in ERα (-) breast cancer cells were tested both in vivo and in vitro.
Results: UCH-L1 expression was conversely correlated with ERα status in breast cancer, and the negative regulatory effect of UCH-L1 on ERα was mediated by the deubiquitinase-mediated stability of EGFR, which suppresses ERα transcription. High expression of UCH-L1 was associated with poor therapeutic response and prognosis in patients with breast cancer. Up-regulation of ERα caused by UCH-L1 inhibition could significantly enhance the efficacy of tamoxifen and fulvestrant in ERα (-) breast cancer both in vivo and in vitro.
Conclusions: Our results reveal an important role of UCH-L1 in modulating ERα status and demonstrate the involvement of UCH-L1-EGFR signaling pathway, suggesting that UCH-L1 may serve as a novel adjuvant target for treatment of hormone therapy-insensitive breast cancers. Targeting UCH-L1 to sensitize ER negative breast cancer to anti-estrogen therapy might represent a new therapeutic strategy that warrants further exploration.
Keywords: Ubiquitin carboxyl terminal hydrolase-L1, Estrogen receptor α, EGFR, ER-negative breast cancer, Endocrine therapy
 Global reach, higher impact
Global reach, higher impact