13.3
Impact Factor
Theranostics 2020; 10(4):1798-1813. doi:10.7150/thno.38210 This issue Cite
Research Paper
Novel ESCC-related gene ZNF750 as potential Prognostic biomarker and inhibits Epithelial-Mesenchymal Transition through directly depressing SNAI1 promoter in ESCC
1. Department of Pathology & Shanxi Key Laboratory of Carcinogenesis and Translational Research on Esophageal Cancer, Shanxi Medical University, Taiyuan, Shanxi 030001 P. R. China
2. College of Letter & Science, University of California Berkeley, Berkeley, California, USA
3. Department of Tumor Surgery, Shanxi Cancer Hospital, Taiyuan, Shanxi 030001, P.R. China
# These authors contributed equally to this work.
Abstract
Background: Cancer genomic studies have identified Zinc Finger Protein 750 (ZNF750) was a novel significantly mutated gene in esophageal squamous cell carcinoma (ESCC). This study was designed to determine the clinical value and molecular mechanisms of ZNF750 in the development of ESCC.
Methods: Genomic data from 4 reported ESCC cohorts were used to analyze the mutation profile of ZNF750. Tissue microarrays were used to detect its expression in 308 ESCC samples. Furtherly, the effects of ZNF750 on proliferation, colony formation, migration and invasion were tested in ESCC cells. PCR-array, chromatin immunoprecipitation (ChIP), luciferase reporter assays, and rescue assay were used to explore the mechanism of ZNF750. Correlation of ZNF750 with its target genes was analyzed in TCGA data from various SCC types.
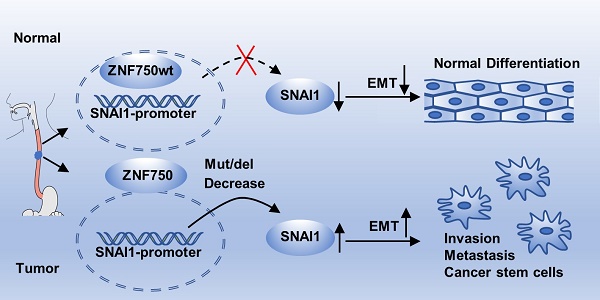
Results: ZNF750 was frequently mutated in ESCC and the most common type was nonsense mutation. Its nucleus/cytoplasm ratio in ESCC was significantly lower than that in paired non-tumor tissues; it was an independent and potential predictor for survival in ESCC patients. Furtherly, ZNF750 knockdown significantly promoted proliferation, colony formation, migration and invasion in ESCC cells. PCR-array showed epithelial-to-mesenchymal transition (EMT) was the main biologic process affected by ZNF750. Moreover, ZNF750 directly bound to the promoter region of SNAI1 and depressed its activity. Decreased ZNF750 up-regulated SNAI1 expression and promoted EMT phenotype. SNAI1 knockdown partially reversed the malignant phenotype induced by ZNF750 knockdown. Further TCGA data analyses showed ZNF750 expression was positively correlated with E-cadherin and negatively correlated with SNAI1, N-cadherin and Vimentin in ESCC and other SCC samples.
Conclusion: Our results suggest that ZNF750 may act as a tumor suppressor by directly repressing SNAI1 and inhibiting EMT process in ESCC and other types of SCC.
Keywords: ESCC, mutation, ZNF750, epithelial-to-mesenchymal transition, SNAI1.
 Global reach, higher impact
Global reach, higher impact