Impact Factor
Theranostics 2020; 10(4):1777-1797. doi:10.7150/thno.36218 This issue Cite
Review
The “ART” of Epigenetics in Melanoma: From histone “Alterations, to Resistance and Therapies”
1. Université Nice Côte d'Azur, Inserm, C3M, France
2. Biology and pathologies of melanocytes, Equipe labellisée ARC 2019, C3M, team 1, France
Abstract
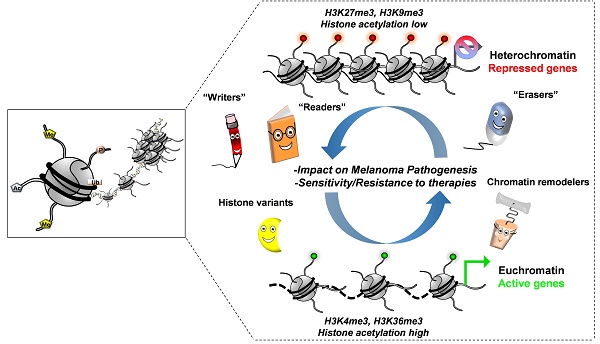
Malignant melanoma is the most deadly form of skin cancer. It originates from melanocytic cells and can also arise at other body sites. Early diagnosis and appropriate medical care offer excellent prognosis with up to 5-year survival rate in more than 95% of all patients. However, long-term survival rate for metastatic melanoma patients remains at only 5%. Indeed, malignant melanoma is known for its notorious resistance to most current therapies and is characterized by both genetic and epigenetic alterations. In cutaneous melanoma (CM), genetic alterations have been implicated in drug resistance, yet the main cause of this resistance seems to be non-genetic in nature with a change in transcription programs within cell subpopulations. This change can adapt and escape targeted therapy and immunotherapy cytotoxic effects favoring relapse.
Because they are reversible in nature, epigenetic changes are a growing focus in cancer research aiming to prevent or revert the drug resistance with current therapies. As such, the field of epigenetic therapeutics is among the most active area of preclinical and clinical research with effects of many classes of epigenetic drugs being investigated. Here, we review the multiplicity of epigenetic alterations, mainly histone alterations and chromatin remodeling in both cutaneous and uveal melanomas, opening opportunities for further research in the field and providing clues to specifically control these modifications. We also discuss how epigenetic dysregulations may be exploited to achieve clinical benefits for the patients, the limitations of these therapies, and recent data exploring this potential through combinatorial epigenetic and traditional therapeutic approaches.
Keywords: melanoma, epigenetics, drug resistance, targeted therapy, immunotherapy
 Global reach, higher impact
Global reach, higher impact