13.3
Impact Factor
Theranostics 2020; 10(4):1678-1693. doi:10.7150/thno.40482 This issue Cite
Research Paper
Development of a new class of PSMA radioligands comprising ibuprofen as an albumin-binding entity
1. Department of Chemistry and Applied Biosciences, ETH Zurich, 8093 Zurich, Switzerland.
2. Center for Radiopharmaceutical Sciences ETH-PSI-USZ, Paul Scherrer Institute, 5232 Villigen-PSI, Switzerland.
3. Isotope Technologies Garching GmbH, 85748 Garching, Germany.
*equally contributed to this manuscript
Abstract
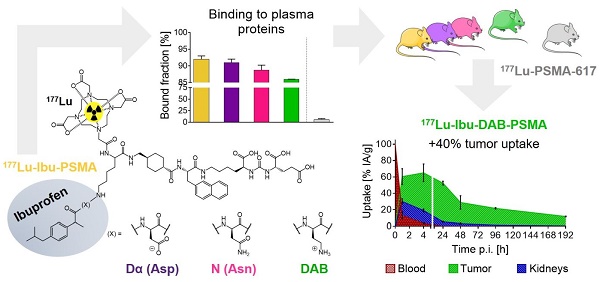
Prostate-specific membrane antigen (PSMA)-targeted radioligands have been used for the treatment of metastatic castration-resistant prostate cancer (mCRPC). Recently, albumin-binding PSMA radioligands with enhanced blood circulation were developed to increase the tumor accumulation of activity. The present study aimed at the design, synthesis and preclinical evaluation of a novel class of PSMA-targeting radioligands equipped with ibuprofen as a weak albumin-binding entity in order to improve the pharmacokinetic properties.
Methods: Four novel glutamate-urea-based PSMA ligands were synthesized with ibuprofen, conjugated via variable amino acid-based linker entities. The albumin-binding properties of the 177Lu-labeled PSMA ligands were tested in vitro using mouse and human plasma. Affinity of the radioligands to PSMA and cellular uptake and internalization was investigated using PSMA-positive PC-3 PIP and PSMA-negative PC-3 flu tumor cells. The tissue distribution profile of the radioligands was assessed in biodistribution and imaging studies using PC-3 PIP/flu tumor-bearing nude mice.
Results: The PSMA ligands were obtained in moderate yields at high purity (>99%). 177Lu-labeling of the ligands was achieved at up to 100 MBq/nmol with >96% radiochemical purity. In vitro assays confirmed high binding of all radioligands to mouse and human plasma proteins and specific uptake and internalization into PSMA-positive PC-3 PIP tumor cells. Biodistribution studies and SPECT/CT scans revealed high accumulation in PC-3 PIP tumors but negligible uptake in PC-3 flu tumor xenografts as well as rapid clearance of activity from background organs and tissues. 177Lu-Ibu-DAB-PSMA, in which ibuprofen was conjugated via a positively-charged diaminobutyric acid (DAB) entity, showed distinguished tumor uptake and the most favorable tumor-to-blood and tumor-to-kidney ratios.
Conclusion: The high accumulation of activity in the tumor and fast clearance from background organs was a common favorable characteristic of PSMA radioligands modified with ibuprofen as albumin-binding entity. 177Lu-Ibu-DAB-PSMA emerged as the most promising candidate; hence, more detailed preclinical investigations with this radioligand are warranted in view of a clinical translation.
Keywords: prostate cancer, PSMA ligands, 177Lu, albumin-binder, ibuprofen.
 Global reach, higher impact
Global reach, higher impact