Impact Factor
Theranostics 2020; 10(4):1604-1618. doi:10.7150/thno.40908 This issue Cite
Research Paper
Investigating the effects of dexamethasone on blood-brain barrier permeability and inflammatory response following focused ultrasound and microbubble exposure
1. Physical Science Platform, Sunnybrook Research Institute, Toronto, ON, Canada.
2. Department of Medical Biophysics, University of Toronto, Toronto, ON, Canada.
3. Institute of Biomaterials and Biomedical Engineering, University of Toronto, Toronto, ON, Canada.
Abstract
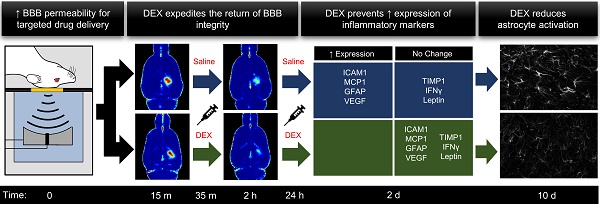
Rationale: Clinical trials are currently underway to test the safety and efficacy of delivering therapeutic agents across the blood-brain barrier (BBB) using focused ultrasound and microbubbles (FUS+MBs). While acoustic feedback control strategies have largely minimized the risk of overt tissue damage, transient induction of inflammatory processes have been observed following sonication in preclinical studies. The goal of this work was to explore the potential of post-sonication dexamethasone (DEX) administration as a means to mitigate treatment risk. Vascular permeability, inflammatory protein expression, blood vessel growth, and astrocyte activation were assessed.
Methods: A single-element focused transducer (transmit frequency = 580 kHz) and DefinityTM microbubbles were used to increase BBB permeability unilaterally in the dorsal hippocampi of adult male rats. Sonicating pressure was calibrated based on ultraharmonic emissions. Dynamic contrast-enhanced magnetic resonance imaging (DCE-MRI) was used to quantitatively assess BBB permeability at 15 min (baseline) and 2 hrs following sonication. DEX was administered following baseline imaging and at 24 hrs post-FUS+MB exposure. Expression of key inflammatory proteins were assessed at 2 days, and astrocyte activation and blood vessel growth were assessed at 10 days post-FUS+MB exposure.
Results: Compared to saline-treated control animals, DEX administration expedited the restoration of BBB integrity at 2 hrs, and significantly limited the production of key inflammation-related proteins at 2 days, following sonication. Indications of FUS+MB-induced astrocyte activation and vascular growth were diminished at 10 days in DEX-treated animals, compared to controls.
Conclusions: These results suggest that DEX provides a means of modulating the duration of BBB permeability enhancement and may reduce the risk of inflammation-induced tissue damage, increasing the safety profile of this drug-delivery strategy. This effect may be especially relevant in scenarios for which the goal of treatment is to restore or preserve neural function and multiple sonications are required.
Keywords: blood-brain barrier, dexamethasone, dynamic contrast-enhanced magnetic resonance imaging, inflammation, focused ultrasound
 Global reach, higher impact
Global reach, higher impact