Impact Factor
Theranostics 2020; 10(4):1514-1530. doi:10.7150/thno.41000 This issue Cite
Research Paper
The Effect of Cardiogenic Factors on Cardiac Mesenchymal Cell Anti-Fibrogenic Paracrine Signaling and Therapeutic Performance
1. Diabetes and Obesity Center, Department of Medicine, University of Louisville, Louisville, KY, USA.
2. The Christina Lee Brown Envirome Institute, Department of Medicine, University of Louisville, Louisville, KY, USA.
3. Institute of Molecular Cardiology, Department of Medicine, University of Louisville, Louisville, KY, USA.
4. Cardiovascular Innovation Institute, Department of Medicine, University of Louisville, Louisville, KY, USA.
*Authors contributed equally to this work.
Abstract
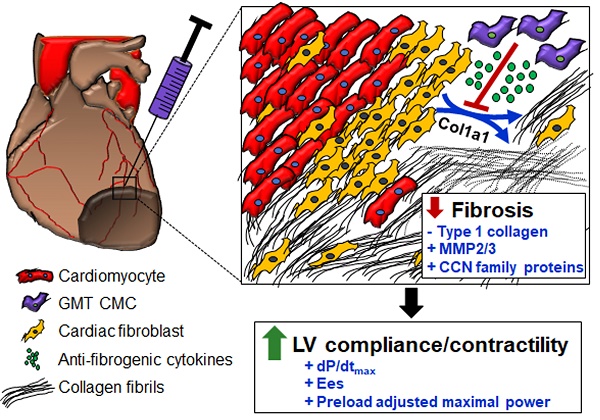
Intrinsic cardiogenic factor expression, a proxy for cardiomyogenic lineage commitment, may be an important determinant of donor cell cardiac reparative capacity in cell therapy applications; however, whether and how this contributes to their salutary effects remain largely ambiguous.
Methods: The current study examined the consequences of enhanced cardiogenic factor expression, via lentiviral delivery of GMT (GATA4, MEF2C, and TBX5), on cardiac mesenchymal cell (CMC) anti-fibrogenic paracrine signaling dynamics, in vitro, and cardiac reparative capacity, in vivo. Proteome cytokine array analyses and in vitro cardiac fibroblast activation assays were performed using conditioned medium derived from either GMT- or GFP control-transduced CMCs, to respectively assess cardiotrophic factor secretion and anti-fibrogenic paracrine signaling aptitude.
Results: Relative to GFP controls, GMT CMCs exhibited enhanced secretion of cytokines implicated to function in pathways associated with matrix remodeling and collagen catabolism, and more ably impeded activated cardiac fibroblast Col1A1 synthesis in vitro. Following their delivery in a rat model of chronic ischemic cardiomyopathy, conventional echocardiography was unable to detect a therapeutic advantage with either CMC population; however, hemodynamic analyses identified a modest, yet calculable supplemental benefit in surrogate measures of global left ventricular contractility with GMT CMCs relative to GFP controls. This phenomenon was neither associated with a decrease in infarct size nor an increase in viable myocardium, but with only a marginal decrease in regional myocardial collagen deposition.
Conclusion: Overall, these results suggest that CMC cardiomyogenic lineage commitment biases cardiac repair and, further, that enhanced anti-fibrogenic paracrine signaling potency may underlie, in part, their improved therapeutic utility.
Keywords: Cardiac Mesenchymal Cells, Therapy, Myocardial Infarction, Fibrosis, Paracrine Signaling
 Global reach, higher impact
Global reach, higher impact