13.3
Impact Factor
Theranostics 2020; 10(25):11562-11579. doi:10.7150/thno.45459 This issue Cite
Research Paper
Extracellular vesicles derived from Krüppel-Like Factor 2-overexpressing endothelial cells attenuate myocardial ischemia-reperfusion injury by preventing Ly6Chigh monocyte recruitment
1. Department of Cardiology, Nanjing Drum Tower Hospital, State Key Laboratory of Pharmaceutical Biotechnology, Medical School of Nanjing University, Nanjing, China.
2. Department of Radiology, Nanjing Drum Tower Hospital, State Key Laboratory of Pharmaceutical Biotechnology, Medical School of Nanjing University, Nanjing, China.
3. Department of Cardiology, Nanjing Drum Tower Hospital Clinical College of Nanjing Medical University, Nanjing, China.
#These authors contributed equally to this work.
Abstract
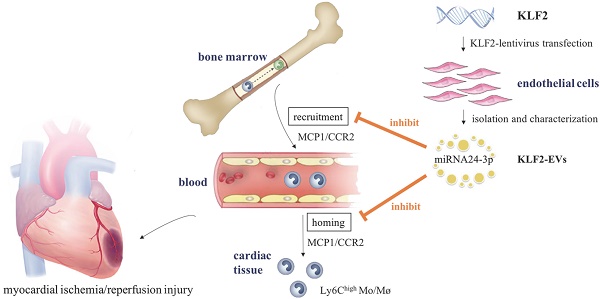
Background: The ischemia/reperfusion (I/R) process in patients with ST-segment elevation myocardial infarction (STEMI) triggers an immune response, resulting in myocyte death. Krüppel-Like Factor 2 (KLF2), which is highly expressed in endothelial cells (ECs) under laminar flow, exerts anti-inflammatory effects. In this study, we explored the role of small extracellular vesicles (EVs) from KLF2-overexpressing ECs (KLF2-EVs) in the immunomodulation and its implications in myocardial I/R injury.
Methods and Results: The small EVs were isolated from KLF2-overexpressing ECs' supernatant using gradient centrifugation. Mice were subjected to 45 min of ischemia followed by reperfusion, and KLF2-EVs were administrated through intravenous injection. KLF2-EVs ameliorated I/R injury and alleviated inflammation level in the serum and heart. We employed the macrophage depletion model and splenectomy and showed that Ly6Chigh monocyte recruitment from bone marrow was the main target of KLF2-EVs. miRNA-sequencing of KLF2-EVs and bioinformatics analysis implicated miRNA-24-3p (miR-24-3p) as a potent candidate mediator of monocyte recruitment and CCR2 as a downstream target. miR-24-3p mimic inhibited the migration of Ly6Chigh monocytes, and miR-24-3p antagomir reversed the effect of KLF2-EVs in myocardial I/R.
Conclusion: Our data demonstrated that KLF2-EVs attenuated myocardial I/R injury in mice via shuttling miR-24-3p that restrained the Ly6Chigh monocyte recruitment. Thus, KLF2-EVs could be a potential therapeutic agent for myocardial I/R injury.
Keywords: Krüppel-Like Factor 2, extracellular vesicles, myocardial ischemia/reperfusion injury
 Global reach, higher impact
Global reach, higher impact