Impact Factor
Theranostics 2019; 9(24):7168-7183. doi:10.7150/thno.36830 This issue Cite
Research Paper
Pancreatic stellate cells activated by mutant KRAS-mediated PAI-1 upregulation foster pancreatic cancer progression via IL-8
1. Clinical Medicine Research Center, National Cheng Kung University Hospital, Tainan, Taiwan.
2. Institute of Clinical Medicine, College of Medicine, National Cheng Kung University, Tainan, Taiwan.
3. Department of Surgery, National Cheng Kung University Hospital, Tainan, Taiwan.
4. Department of Medical Oncology, Kaohsiung Chang Gung Memorial Hospital, Kaohsiung, Taiwan.
5. Graduate Institute of Clinical Medical Sciences, Chang Gung University College of Medicine, Kaohsiung, Taiwan.
6. Department of Biochemistry and Molecular Biology, College of Medicine, National Cheng Kung University, Tainan, Taiwan.
7. International Research Center of Wound Regeneration & Repair, National Cheng Kung University, Tainan, Taiwan.
8. Department of Physiology, College of Medicine, National Cheng Kung University Tainan, Taiwan.
9. National Institute of Cancer Research, National Healthy Research Institute, Tainan, Taiwan.
*Equal contribution to this study
Abstract
Background: The dense fibrotic stroma enveloping pancreatic tumors is a major cause of drug resistance. Pancreatic stellate cells (PSCs) in the stroma can be activated to induce intra-tumor fibrosis and worsen patient survival; however, the molecular basics for the regulation of PSC activation remains unclear.
Methods: The in vitro coculture system was used to study cancer cell-PSC interactions. Atomic force microscopy was used to measure the stiffness of tumor tissues and coculture gels. Cytokine arrays, qPCR, and Western blotting were performed to identify the potential factors involved in PSC activation and to elucidate underlying pathways.
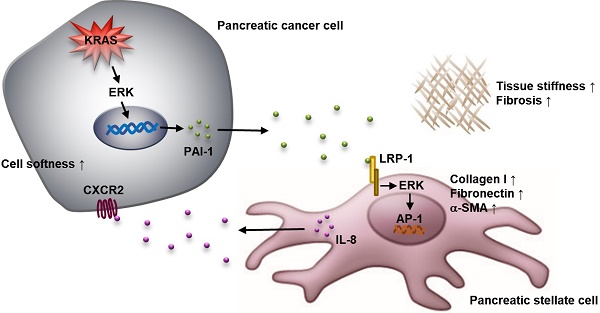
Results: PSC activation characterized by α-SMA expression was associated with increased pancreatic tumor stiffness and poor prognosis. Coculture with cancer cells induced PSC activation, which increased organotypic coculture gel stiffness and cancer cell invasion. Cancer cells-derived PAI-1 identified from coculture medium could activate PSCs, consistent with pancreatic cancer tissue microarray analysis showing a strong positive correlation between PAI-1 and α-SMA expression. Suppression by knocking down PAI-1 in cancer cells demonstrated the requirement of PAI-1 for coculture-induced PSC activation and gel stiffness. PAI-1 could be upregulated by KRAS in pancreatic cancer cells through ERK. In PSCs, inhibition of LRP-1, ERK, and c-JUN neutralized the effect of PAI-1, suggesting the contribution of LRP-1/ERK/c-JUN signaling. Furthermore, activated PSCs might exacerbate malignant behavior of cancer cells via IL-8 because suppression of IL-8 signaling reduced pancreatic tumor growth and fibrosis in vivo.
Conclusions: KRAS-mutant pancreatic cancer cells can activate PSCs through PAI-1/LRP-1 signaling to promote fibrosis and cancer progression.
Keywords: pancreatic cancer, PSC, stiffness, organotypic coculture, PAI-1
 Global reach, higher impact
Global reach, higher impact