13.3
Impact Factor
Theranostics 2019; 9(20):5937-5955. doi:10.7150/thno.34168 This issue Cite
Research Paper
GPR124 facilitates pericyte polarization and migration by regulating the formation of filopodia during ischemic injury
1. Institute of Pharmacology and Toxicology, College of Pharmaceutical Sciences, Zhejiang University, Hangzhou, 310058, China
2. Key Laboratory of Cardiovascular & Cerebrovascular Medicine, School of Pharmacy, Nanjing Medical University, Nanjing, 211166, China
3. Department of Physiology, Nanjing Medical University, Nanjing, 211166, China
4. School of Medicine, Zhejiang University City College, Hangzhou, 310015, China
5. Departments of Pharmaceutical, Administrative, and Basic Sciences, Schools of Pharmacy and Medicine, Loma Linda University Health, CA, 92350, USA
6. Department of Pharmacology, Graduate School of Pharmaceutical Sciences, Tohoku University, Sendai, 980-8578, Japan
*These authors contributed equally to this work.
Abstract
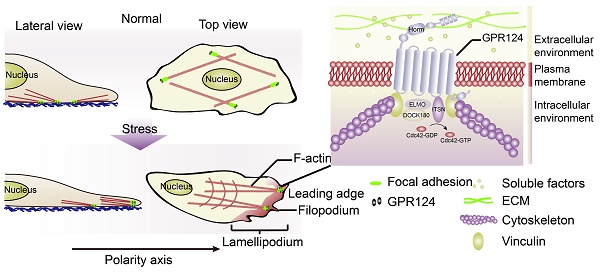
Prolonged occlusion of multiple microvessels causes microvascular injury. G protein-coupled receptor 124 (GPR124) has been reported to be required for maintaining central nervous system (CNS) angiogenesis and blood-brain barrier integrity. However, the molecular mechanisms by which GPR124 regulates pericytes during ischemia have remained elusive.
Methods: A microsphere embolism-induced ischemia model was used to evaluate the expression of GPR124 following microsphere embolism. Immunocytochemistry and stochastic optical reconstruction microscopy imaging were used to assess the expression and distribution of GPR124 in human brain vascular pericytes (HBVPs) and after the treatment with 3-morpholino-sydnonimine (SIN-1) or oxygen-glucose deprivation (OGD). The effect of GPR124 knockdown or overexpression on HBVP migration was analyzed in vitro using wound healing assays and a microfluidic device. GPR124 loss-of-function studies were performed in HBVPs and HEK293 cells using CRISPR-Cas9-mediated gene deletion. Time-lapse imaging was used to assess dynamic changes in the formation of filopodia in an individual cell. Finally, to explore the functional domains required for GPR124 activity, deletion mutants were constructed for each of the N-terminal domains.
Results: GPR124 expression was increased in pericytes following microsphere embolism. Morphological analysis showed localization of GPR124 to focal adhesions where GPR124 bound directly to the actin binding protein vinculin and upregulated Cdc42. SIN-1 or OGD treatment redistributed GPR124 to the leading edges of HBVPs where GPR124 signaling was required for pericyte filopodia formation and directional migration. Partial deletion of GPR124 domains decreased SIN-1-induced filopodia formation and cell migration.
Conclusion: Taken together, our results provide the first evidence for a role of GPR124 in pericyte migration under ischemic conditions and suggest that GPR124 was essential for Cdc42 activation and filopodia formation.
Keywords: GPR124, focal adhesions, pericytes, directional migration, filopodia, ischemia.
 Global reach, higher impact
Global reach, higher impact