13.3
Impact Factor
Theranostics 2019; 9(20):5797-5809. doi:10.7150/thno.35059 This issue Cite
Research Paper
Glyco-Dendrimers as Intradermal Anti-Tumor Vaccine Targeting Multiple Skin DC Subsets
1. Amsterdam UMC, Department of Molecular Cell Biology and Immunology, Cancer Center Amsterdam, Amsterdam Infection and Immunity Institute, Vrije Universiteit Amsterdam, Amsterdam, Netherlands
2. Amsterdam UMC, Department of Medical Oncology, Cancer Center Amsterdam, Vrije Universiteit Amsterdam, Amsterdam, Netherlands
Abstract
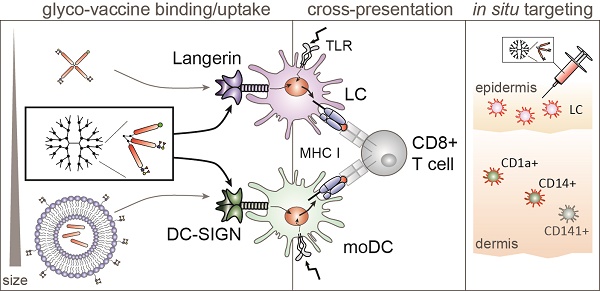
The human skin is an attractive anti-tumor vaccination site due to the vast network of dendritic cell (DC) subsets that carry antigens to the draining lymph nodes and stimulate tumor specific CD4+ and CD8+ T cells in. Specific vaccine delivery to skin DC can be accomplished by targeting glycan coated antigens to C-type lectin receptors (CLRs) such as DC-SIGN expressed by human dermal DCs and Langerin expressed by Langerhans cells (LCs), which facilitate endocytosis and processing for antigen presentation and T cell activation. Although there are multiple human skin DC subsets, targeting individual DC subsets and receptors has been a focus in the past. However, the simultaneous targeting of multiple human skin DC subsets that mobilize the majority of the skin antigen presenting cells (APC) is preferred to accomplish more robust and efficient T cell stimulation. Dual CLR targeting using a single tumor vaccine has been difficult, as we previously showed Langerin to favor binding and uptake of monovalent glyco-peptides whereas DC-SIGN favors binding of larger multivalent glyco-particles such as glyco-liposomes.
Methods: We used branched polyamidoamine (PAMAM) dendrimers as scaffold for melanoma specific gp100 synthetic long peptides and the common DC-SIGN and Langerin ligand Lewis Y (LeY), to create multivalent glyco-dendrimers with varying molecular weights for investigating dual DC-SIGN and Langerin targeting. Using DC-SIGN+ monocyte derived DC (moDC) and Langerin+ primary LC we investigated glyco-dendrimer CLR targeting properties and subsequent gp100 specific CD8+ T cell activation in vitro. In situ targeting ability to human dermal DC and LC through intradermal injection in a human skin explant model was elucidated.
Results: Dual DC-SIGN and Langerin binding was achieved using glyco-dendrimers of approximately 100kD, thereby fulfilling our criteria to simultaneously target LCs and CD1a+ and CD14+ dermal DC in situ. Both DC-SIGN and Langerin targeting by glyco-dendrimers resulted in enhanced internalization and gp100 specific CD8+ T cell activation.
Conclusion: We designed the first glyco-vaccine with dual CLR targeting properties, thereby reaching multiple human skin DC subsets in situ for improved anti-tumor CD8+ T cell responses.
Keywords: DC-SIGN, Langerin, glyco-dendrimer, intradermal vaccination, tumor
 Global reach, higher impact
Global reach, higher impact