Impact Factor
Theranostics 2019; 9(20):5769-5783. doi:10.7150/thno.36505 This issue Cite
Research Paper
Inhibition of HSP90β Improves Lipid Disorders by Promoting Mature SREBPs Degradation via the Ubiquitin-proteasome System
1. State Key Laboratory of Natural Medicines, China Pharmaceutical University, 210009, Nanjing, Jiangsu, China.
2. Jiangsu Key Laboratory of Drug Discovery for Metabolic Diseases, China Pharmaceutical University, 210009, Nanjing, Jiangsu, China.
3. Shanghai Diabetes Institute, Department of Endocrinology and Metabolism, Shanghai Jiao Tong University Affiliated Sixth People's Hospital, 200233, Shanghai, China.
4. Department of General Practice and Lab of PTM, State Key Laboratory of Biotherapy, West China Hospital, Sichuan University, and Collaborative Innovation Center of Biotherapy, Chengdu 610041, China.
5. Institute of Dermatology, Chinese Academy of Medical Sciences, Peking Union Medical College. 210009, Nanjing, Jiangsu, China.
6. Department of Infection Diseases, Hangzhou First People's Hospital, 310006, Zhejiang, China.
7. State Key Laboratory of Pharmaceutical Biotechnology and Jiangsu Key Laboratory of Molecular Medicine and School of Medicine, Nanjing University, 210093, Nanjing, Jiangsu, China.
8. Key Laboratory of Nutrition and Metabolism, Institute for Nutritional Sciences, Shanghai Institutes for Biological Sciences, Chinese Academy of Sciences, University of Chinese Academy of Sciences, 200031, Shanghai, China.
9. School of Pharmaceutical Sciences, Tsinghua University, Beijing, 100084.
10. Drug Discovery and Design Center, CAS Key Laboratory of Receptor Research, Shanghai Institute of Materia Medica, Chinese Academy of Sciences, 555 Zuchongzhi Road, Shanghai, 201203, China.
11. Department of Hepatobiliary Surgery, Xi-Jing Hospital, Fourth Military Medical University, Xi'an, 710032, China.
Abstract
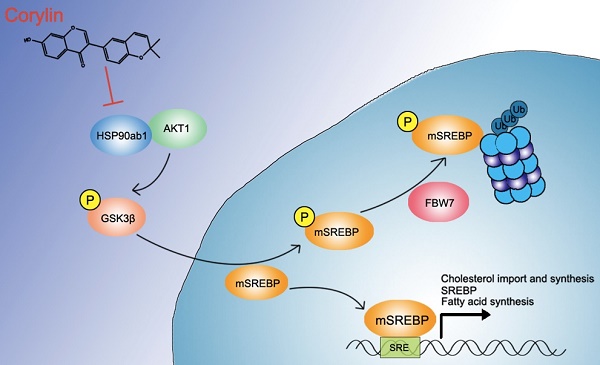
Rationale: Heat shock protein 9 (HSP90) are a family of the most highly expressed cellular proteins and attractive drug targets against cancer, neurodegeneration diseases, etc. HSP90 proteins have also been suggested to be linked to lipid metabolism. However, the specific function of HSP90 paralogs, as well as the underlying molecular cascades remains largely unknown. This study aims to unravel the paralog-specific role of HSP90 in lipid metabolism and try to discover paralog-specific HSP90 inhibitors.
Methods: In non-alcohol fatty liver disease (NAFLD) patients, as well as in diet induced obese (DIO) mice, expression of HSP90 paralogs were analyzed by immunohistochemistry and western blot. In hepatocytes and in DIO mice, HSP90 proteins were knockdown by siRNAs/shRNAs, metabolic parameters, as well as downstream signaling were then investigated. By virtue screening, corylin was found to bind specifically to HSP90β. Using photo-affinity labeling and mass spectrum, corylin binding proteins were identified. After oral administration of corylin, its lipid lowering effects in different metabolic disease mice models were evaluated.
Results: We showed that hepatic HSP90β, rather than HSP90α, was overexpressed in NAFLD patients and obese mice. Hepatic HSP90β was also clinical relevant to serum lipid level. Depletion of HSP90β promoted mature sterol regulatory element-binding proteins (mSREBPs) degradation through Akt-GSK3β-FBW7 pathway, thereby dramatically decreased the content of neutral lipids and cholesterol. We discovered an HSP90β-selective inhibitor (corylin) that only bound to its middle domain. We found that corylin treatment partially suppressed Akt activity only at Thr308 site and specifically promoted mSREBPs ubiquitination and proteasomal degradation. Corylin treatment significantly reduced lipid content in both liver cell lines and human primary hepatocytes. In animal studies, we showed that corylin ameliorated obesity-induced fatty liver disease, type 2 diabetes and atherosclerosis.
Principle conclusions: HSP90β plays a parolog-specific role in regulating lipid homeostasis. Compound that selectively inhibits HSP90β could be useful in the clinic for the treatment for metabolic diseases.
Keywords: HSP90β, lipid metabolism, corylin, SREBPs, proteasomal degradation
 Global reach, higher impact
Global reach, higher impact