13.3
Impact Factor
Theranostics 2019; 9(20):5739-5754. doi:10.7150/thno.37556 This issue Cite
Research Paper
Decreased expression of mitochondrial miR-5787 contributes to chemoresistance by reprogramming glucose metabolism and inhibiting MT-CO3 translation
1. Guangdong Provincial Key Laboratory of Malignant Tumor Epigenetics and Gene Regulation of Sun Yat-Sen Memorial Hospital, Guangzhou 510120, China
2. Department of Oral and Maxillofacial Surgery, Sun Yat-Sen Memorial Hospital of Sun Yat-Sen University, Guangzhou 510120, China
3. Division of Nephrology, Nanfang Hospital, Southern Medical University, State Key Laboratory of Organ Failure Research, National Clinical Research Center of Kidney Disease, Guangzhou 510515, China.
4. State University of New York at Stony Brook, Stony Brook, NY, 11794, USA
5. Markey Cancer Center, the University of Kentucky, College of Medicine, Lexington, Kentucky 40506, USA
6. Nanchang Key Laboratory of Cancer Pathogenesis and Translational Research, Center Laboratory, the Third Affiliated Hospital, Nanchang University, Nanchang 330047, China
7. Massachusetts General Hospital Cancer Center, Harvard Medical School, Boston, MA02114, USA
8. Department of Surgery, Massachusetts General Hospital, Harvard Medical School, 55 Fruit Street, Boston, MA 02114, USA
9. Experimental Therapeutics and Molecular Imaging Lab, Department of Neurology, Massachusetts General Hospital and Harvard Medical School, Boston, MA 02129, USA
*These authors contributed equally to this work.
Abstract
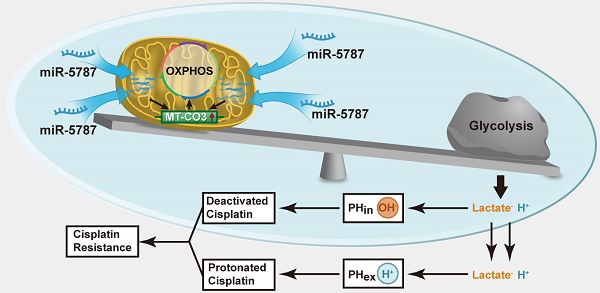
MicroRNAs (miRNAs) have been recently found in the mitochondria, and were named “mitomiRs”, but their function has remained elusive. Here, we aimed to assess the presence and function(s) of mitomiRs in tongue squamous cell carcinoma (TSCC).
Methods: miRNA microarray was performed in paired TSCC cell lines, Cal27 and its chemoresistant counterpart, Cal27-re. Decreased expression of mitomiRs in chemoresistant cells was characterized. The functions of mitomiRs were investigated by a series of in vitro and in vivo experiments.
Results: Differential microarray analysis identified downregulation of mitomiR-5787 in Cal27-re cells. We knocked down mitomiR-5787 in parental cells and upregulated its expression in cisplatin-resistant cells. The sensitivity of TSCC cells to cisplatin was regulated by miR-5787. The glucose metabolism assay suggested that reduced expression of miR-5787 changed the balance of glucose metabolism by shifting it from oxidative phosphorylation to aerobic glycolysis. Xenograft experiments in BALB/c-nu mice further verified the in vitro results. Reduced expression of miR-5787 contributes to chemoresistance in TSCC cells by inhibiting the translation of mitochondrial cytochrome c oxidase subunit 3 (MT-CO3). The prognostic analysis of 126 TSCC patients showed that the patients with low expression of miR-5787 and/or MT-CO3 had poor cisplatin sensitivity and prognosis.
Conclusions: Mitochondrial miR-5787 could regulate cisplatin resistance of TSCC cells and affect oxidative phosphorylation and aerobic glycolysis. Downregulation of miR-5787 inhibited the translation of MT-CO3 to regulate cisplatin resistance of TSCC. Mitochondrial miR-5787 and MT-CO3 can be used as predictive biomarkers or therapeutic targets for cisplatin chemotherapy resistance.
Keywords: mitochondrial miR-5787, oxidative phosphorylation, aerobic glycolysis, cisplatin chemotherapy resistance, MT-CO3, tongue squamous cell carcinoma
 Global reach, higher impact
Global reach, higher impact