Impact Factor
Theranostics 2019; 9(20):5731-5738. doi:10.7150/thno.34402 This issue Cite
Research Paper
A Theranostic Cathepsin Activity-Based Probe for Noninvasive Intervention in Cardiovascular Diseases
1. Institute for Drug Research, School of Pharmacy, Faculty of Medicine, The Hebrew University, Jerusalem, Israel, 9112001.
2. Heart institute, Hadassah, University Hospital, Jerusalem, Israel 91120.
*These authors contributed equally to this work.
Abstract
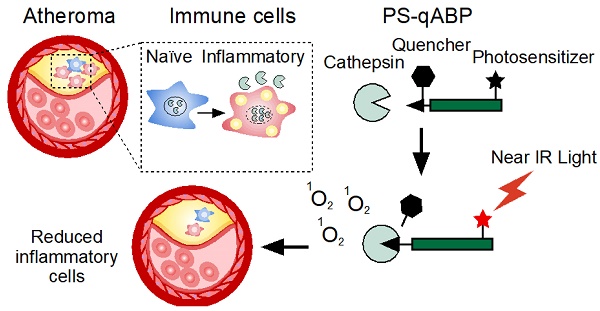
Despite the common use of lipid-lowering medications, cardiovascular diseases continue to be a significant health concern. Atherosclerosis, one of the most frequent causes of cardiovascular morbidity, involves extensive inflammatory activity and remodeling of the vascular endothelium. This relentless inflammatory condition can ultimately give rise to clinical manifestations, such as ischemic heart disease or stroke. Accumulating evidence over the past decades implicates cysteine protease cathepsins in cardiovascular disorders. In particular, Cathepsins B, L, and S are over-expressed during vascular inflammation, and their activity is associated with impaired clinical outcomes. Here we took advantage of these molecular events to introduce a non-invasive detection and treatment approach to modulate vascular inflammation using a Photosensitizing quenched Activity-Based Probed (PS-qABP) that targets these proteases.
Methods: We tested the application of this approach in LDL receptor-deficient mice and used non-invasive imaging and heart cross-section staining to assess the theranostic efficacy of this probe. Moreover, we used fresh human endarterectomy tissues to analyze cathepsin signals on gel, and verified cathepsin identity by mass spectrometry.
Results: We showed that our PS-qABP can rapidly accumulate in areas of inflammatory atheromas in vivo, and application of light therapy profoundly reduced lesional immune cell content without affecting smooth muscle cell and collagen contents. Lastly, using human tissue samples we provided proof-of-concept for future clinical applications of this technology.
Conclusions: Photodynamic therapy guided by cysteine cathepsin activity is an effective approach to reduce vascular inflammation and attenuate atherosclerosis progression. This approach could potentially be applied in clinical settings.
Keywords: cathepsins, macrophages, photodynamic therapy, activity-based probes, atherosclerosis
 Global reach, higher impact
Global reach, higher impact