13.3
Impact Factor
Theranostics 2019; 9(10):2882-2896. doi:10.7150/thno.32693 This issue Cite
Research Paper
The cell-penetrating FOXM1 N-terminus (M1-138) demonstrates potent inhibitory effects on cancer cells by targeting FOXM1 and FOXM1-interacting factor SMAD3
State Key Laboratory of Chemo/Biosensing and Chemometrics, College of Biology, Hunan Engineering Research Center for Anticancer Targeted Protein Pharmaceuticals, Hunan University, Changsha, Hunan 410082, China.
Abstract
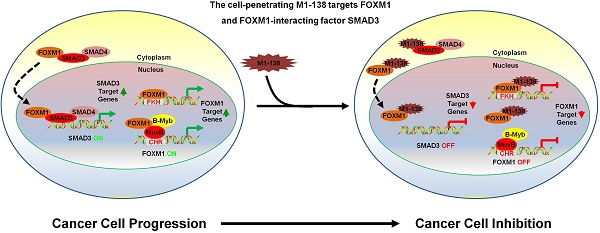
Transcription factor FOXM1 is involved in stimulating cell proliferation, enhancing DNA damage repair, promoting metastasis of cancer cells, and the inhibition of FOXM1 has been shown to prevent the initiation and progression of multiple cancers and FOXM1 is considered to be an effective target for tumor therapeutic drug development. The N-terminus of FOXM1 has been found to prevent transcriptional activities of FOXM1 and to mediate the interaction between FOXM1 and SMAD3.
Methods: A recombinant FOXM1 N-terminal domain (1-138aa) fused with a nine arginine cell-penetrating peptide is produced with an E. coli expression system and named as M1-138. The effects of M1-138 on the proliferation, migration, and tumorigenic ability of cancer cells are analyzed in vitro with cell counting, transwell assays, and colony formation assays. Electrophoretic mobility shift assays (EMSAs) and Luciferase activity assays are used to test the DNA binding ability and transcriptional activity of transcription factors. The levels of mRNAs and proteins are measured by quantitative-PCR, Western blotting or Immunohistochemistry. The interactions among proteins are analyzed with Pull-down and Co-immunoprecipitation (Co-IP) assays. The nude mouse engrafted tumor models are used to test the inhibitory effects of M1-138 in vivo.
Results: M1-138 diminishes the proliferation and migration abilities of cancer cells through binding to FOXM1 and FOXM1-interacting factor SMAD3, and consequently attenuating FOXM1 transcriptional activities from both direct and indirect FOXM1-promoter binding mechanisms and interfering with the interaction between FOXM1 and SMAD3. Treatment of M1-138 prevents tumorigenicity of cancer cells and inhibits tumor growth in nude mouse xenograft models with no obvious signs of toxicity.
Conclusion: M1-138 is a promising drug candidate for the development of anti-cancer therapeutics targeting FOXM1 and SMAD3.
Keywords: Anticancer protein therapeutics, Transcription factor FOXM1, Targets for tumor suppression, Cell-penetrating peptide
 Global reach, higher impact
Global reach, higher impact