Impact Factor
Theranostics 2019; 9(6):1714-1727. doi:10.7150/thno.30716 This issue Cite
Research Paper
Exosomes from M1-Polarized Macrophages Enhance Paclitaxel Antitumor Activity by Activating Macrophages-Mediated Inflammation
1. School of Pharmacy, Anhui University of Chinese Medicine, Hefei, Anhui, 230012, China
2. Institute of Drug Metabolism, Anhui University of Chinese Medicine, Hefei, Anhui, 230012, China
3. Anhui Province Key Laboratory of Chinese Medicinal Formula, Hefei, Anhui, 230012, China
4. Institute of Pharmaceutics, Anhui Academy of Chinese Medicine, Hefei, Anhui, 230012, China
5. Synergetic Innovation Center of Anhui Authentic Chinese Medicine Quality Improvement, Hefei, Anhui, 230012, China
Abstract
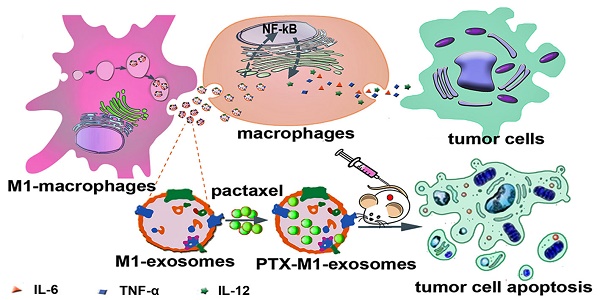
Objective: Exosomes (Exos) are membrane-encased vesicles derived by nearly all cell types for intercellular communication and regulation. They also received attention for their use as natural therapeutic platforms and drug delivery system. Classically activated M1 macrophages suppress tumor growth by releasing pro-inflammatory factors. This study investigated the suitability of M1-exosomes (M1-Exos) as drug carrier and their effect on the NF-κB signal pathway and further detected whether macrophages repolarization can potentiate the antitumor activities of chemotherapeutics.
Methods: M1-Exos were isolated from M1-macrophages by ultracentrifugation and characterized by transmission electron, nanoparticle tracking analysis, dynamic light scattering and western blot. Then M1-Exos were used as Paclitaxel (PTX) carriers to prepare a nano-formulation (PTX- M1-Exos). A relatively simple slight sonication method was used to prepare the drug delivery system (PTX-M1-Exos). The cytotoxicity of PTX-M1-Exos on cancer cells was detected by MTT and flow cytometry in vitro. 4T1 tumor bearing mice were used to perform the therapeutic effect of PTX-M1-Exos in vivo.
Results: The expression of caspase-3 in breast cancer cells was increased when co-incubated with macrophages in the presence of M1-Exos in vitro. The production of pro-inflammatory cytokines was increased after exposure of macrophages in M1-Exos. M1-Exos provided a pro-inflammatory environment which enhanced the anti-tumor activity via caspase-3 mediated pathway. The treatment of M1-Exos to the tumor bearing mice exhibit anti-tumor effects in vivo. Meanwhile, the treatment of PTX-M1-Exos demonstrated higher anti-tumor effects than the M1-Exos or PTX group.
Conclusion: The results in our study indicate that the M1-Exos act as the carrier to deliver PTX into the tumor tissues, and also enhance the anti-tumor effects of chemotherapeutics in tumor bearing mice.
Keywords: anti-cancer, immunological-chemo therapy, M1-macrophage, M1- exosomes, drug delivery
 Global reach, higher impact
Global reach, higher impact