13.3
Impact Factor
Theranostics 2019; 9(6):1614-1633. doi:10.7150/thno.30398 This issue Cite
Research Paper
SIRT3-Dependent Mitochondrial Dynamics Remodeling Contributes to Oxidative Stress-Induced Melanocyte Degeneration in Vitiligo
Department of Dermatology, Xijing hospital, Fourth Military Medical University, Xi'an, Shannxi, China
*These authors contributed equally to this work.
Abstract
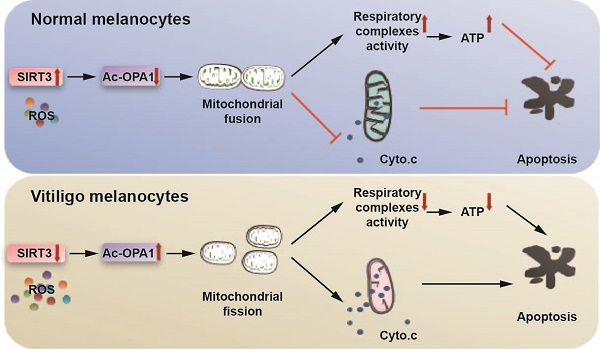
Mitochondrial dysregulation has been implicated in oxidative stress-induced melanocyte destruction in vitiligo. However, the molecular mechanism underlying this process is merely investigated. Given the prominent role of nicotinamide adenine dinucleotide (NAD+)-dependent deacetylase Sirtuin3 (SIRT3) in sustaining mitochondrial dynamics and homeostasis and that SIRT3 expression and activity can be influenced by oxidative stress-related signaling, we wondered whether SIRT3 could play an important role in vitiligo melanocyte degeneration by regulating mitochondrial dynamics.
Methods: We initially testified SIRT3 expression and activity in normal and vitiligo melanocytes via PCR, immunoblotting and immunofluorescence assays. Then, cell apoptosis, mitochondrial function and mitochondrial dynamics after SIRT3 intervention were analyzed by flow cytometry, immunoblotting, confocal laser microscopy, transmission electron microscopy and oxphos activity assays. Chromatin immunoprecipitation (ChIP), co-immunoprecipitation (Co-IP), immunoblotting and immunofluorescence assays were performed to clarify the upstream regulatory mechanism of SIRT3. Finally, the effect of honokiol on protecting melanocytes and the underlying mechanism were investigated via flow cytometry and immunoblotting analysis.
Results: We first found that the expression and the activity of SIRT3 were significantly impaired in vitiligo melanocytes both in vitro and in vivo. Then, SIRT3 deficiency led to more melanocyte apoptosis by inducing severe mitochondrial dysfunction and cytochrome c release to cytoplasm, with Optic atrophy 1 (OPA1)-mediated mitochondrial dynamics remodeling involved in. Moreover, potentiated carbonylation and dampened peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC1α) activation accounted for SIRT3 dysregulation in vitiligo melanocytes. Finally, we proved that honokiol could prevent melanocyte apoptosis under oxidative stress by activating SIRT3-OPA1 axis.
Conclusions: Overall, we demonstrate that SIRT3-dependent mitochondrial dynamics remodeling contributes to oxidative stress-induced melanocyte degeneration in vitiligo, and honokiol is promising in preventing oxidative stress-induced vitiligo melanocyte apoptosis.
Keywords: vitiligo, SIRT3, mitochondrial dynamics, melanocyte, apoptosis
 Global reach, higher impact
Global reach, higher impact