Impact Factor
Theranostics 2018; 8(20):5758-5771. doi:10.7150/thno.28944 This issue Cite
Research Paper
Virtual stenting with simplex mesh and mechanical contact analysis for real-time planning of thoracic endovascular aortic repair
1. School of Life Science, Beijing Institute of Technology, Beijing 100081, China
2. Department of Interventional Neuroradiology, Beijing Tiantan Hospital, Capital Medical University, Beijing 100050, China
3. Institute of Orthopedics, Chinese PLA General Hospital, Beijing 100083, China
4. Department of Diagnostic and Interventional Radiology, Beijing Anzhen Hospital, Capital Medical University, Beijing 100029, China
5. Department of Vascular Surgery, The Second People's Hospital of Yunnan Province, Kunming 650021, China
6. Department of Vascular and Endovascular Surgery, Chinese PLA General Hospital, Beijing 100083, China
Abstract
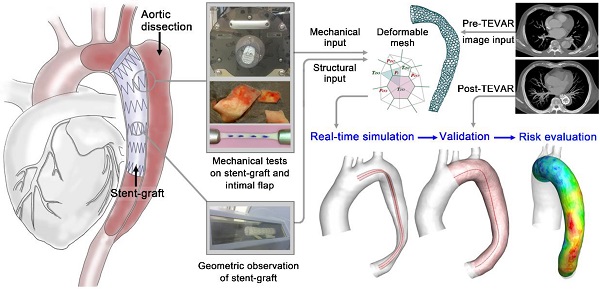
In aortic endovascular repair, the prediction of stented vessel remodeling informs treatment plans and risk evaluation; however, there are no highly accurate and efficient methods to quantitatively simulate stented vessels. This study developed a fast virtual stenting algorithm to simulate stent-induced aortic remodeling to assist in real-time thoracic endovascular aortic repair planning.
Methods: The virtual stenting algorithm was established based on simplex deformable mesh and mechanical contact analysis. The key parameters of the mechanical contact analysis were derived from mechanical tests on aortic tissue (n=40) and commonly used stent-grafts (n=6). Genetic algorithm was applied to select weighting parameters. Testing and validation of the algorithm were performed using pre- and post-treatment computed tomography angiography datasets of type-B aortic dissection cases (n=66).
Results: The algorithm was efficient in simulating stent-induced aortic deformation (mean computing time on a single processor: 13.78±2.80s) and accurate at the morphological (curvature difference: 1.57±0.57%; cross-sectional area difference: 4.11±0.85%) and hemodynamic (similarity of wall shear stress-derived parameters: 90.16-90.94%) levels. Stent-induced wall deformation was higher (p<0.05) in distal stent-induced new entry cases than in successfully treated cases, and this deformation did not differ significantly among the different stent groups. Additionally, the high stent-induced wall deformation regions and the new-entry sites overlapped, indicating the usefulness of wall deformation to evaluate the risks of device-induced complications.
Conclusion: The novel algorithm provided fast real-time and accurate predictions of stent-graft deployment with luminal deformation tracking, thereby potentially informing individualized stenting planning and improving endovascular aortic repair outcomes. Large, multicenter studies are warranted to extend the algorithm validation and determine stress-induced wall deformation cutoff values for the risk stratification of particular complications.
Keywords: Virtual stenting, Simplex deformable mesh, Mechanical analysis, Thoracic endovascular aortic repair, Virtual angiography
 Global reach, higher impact
Global reach, higher impact