13.3
Impact Factor
Theranostics 2018; 8(20):5662-5675. doi:10.7150/thno.28241 This issue Cite
Research Paper
In vivo Imaging-Guided Nanoplatform for Tumor Targeting Delivery and Combined Chemo-, Gene- and Photothermal Therapy
1. Britton Chance Center for Biomedical Photonics at Wuhan National Laboratory for Optoelectronics-Hubei Bioinformatics & Molecular Imaging Key Laboratory, Department of Biomedical Engineering, College of Life Science and Technology, Huazhong University of Science and Technology, Wuhan 430074, Hubei, P. R. China
2. Institute of Medical Engineering, Department of Biophysics, School of Basic Medical Sciences, Xi'an Jiaotong University Health Science Center, Xi'an, Shaanxi, 710061, China
3. Digestive Disease Research Group (DDRG), Institute for Biomedical Sciences, Center for Diagnostics and Therapeutics, Georgia State University, Atlanta, GA, 30302, USA.
4. Key Laboratory of Biomedical Photonics (HUST), Ministry of Education, Huazhong University of Science and Technology, Wuhan 430074, Hubei, P. R. China
*These authors contributed equally to this article.
Abstract
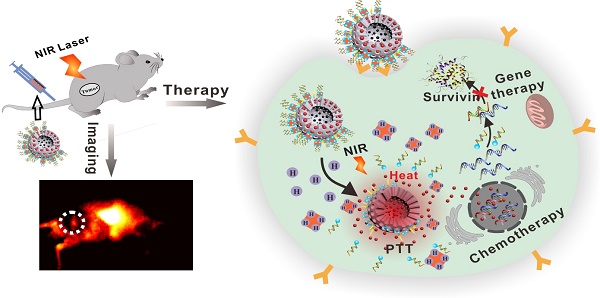
Currently, a large number of anti-tumor drug delivery systems have been widely used in cancer therapy. However, due to the molecular complexity and multidrug resistance of tumors, monotherapies remain suboptimal. Thus, this study aimed to develop a multifunctional theranostic nanoplatform for effective cancer therapy.
Methods: Folic acid-modified silver sulfide@mesoporous silica core-shell nanoparticle was first modified with desthiobiotin (db) on the surface, then doxorubicin (DOX) was loaded into pore. Avidin was employed as "gatekeeper" to prevent leakage of DOX via desthiobiotin-avidin interaction. Db-modified survivin antisense oligonucleotide (db-DNA) which could inhibit survivin expression was then grafted on avidin at the outer layer of nanoparticle. DOX release and db-DNA dissociation were simultaneously triggered by overexpressing biotin in cancer cells, then combining PTT from Ag2S QD to inhibit tumor growth.
Results: This nanoprobe had satisfactory stability and photothermal conversion efficiency up to 33.86% which was suitable for PTT. Due to the good targeting ability and fluorescent anti-bleaching, its signal still existed at the tumor site after tail vein injection of probe into HeLa tumor-bearing nude mice for 48 h. In vitro and in vivo antitumor experiments both demonstrated that drug, gene and photothermal synergistic therapy significantly enhanced antitumor efficacy with minimal systemic toxicity.
Conclusion: Our findings demonstrate that this novel nanoplatform for targeted image-guided treatment of tumor and tactfully integrated chemotherapy, photothermal therapy (PTT) and gene therapy might provide an insight for cancer theranostics.
Keywords: mesoporous silica, fluorescence imaging, photothermal therapy, gene therapy, drug delivery system
 Global reach, higher impact
Global reach, higher impact