Impact Factor
Theranostics 2018; 8(20):5645-5659. doi:10.7150/thno.29840 This issue Cite
Research Paper
In vivo [64Cu]CuCl2 PET imaging reveals activity of Dextran-Catechin on tumor copper homeostasis
1. The Australian Nuclear Science and Technology Organisation, Lucas Heights, NSW, Australia 2234.
2. The Brain and Mind Centre, University of Sydney, Camperdown, NSW, Australia 2050.
3. Children's Cancer Institute, Lowy Cancer Research Centre, UNSW Sydney, NSW, Australia 2031.
4. Università della Calabria, Rende (CS), Italy 87036.
5. The Children's Hospital at Westmead, Westmead, NSW, Australia 2145.
6. ARC Centre of Excellence for Convergent BioNano Science and Technology, Australian Centre for NanoMedicine, UNSW Sydney, NSW, Australia 2052.
7. School of Women's and Children's Health, Faculty of Medicine, UNSW Sydney, NSW, Australia 2052.
*equal contribution
Abstract
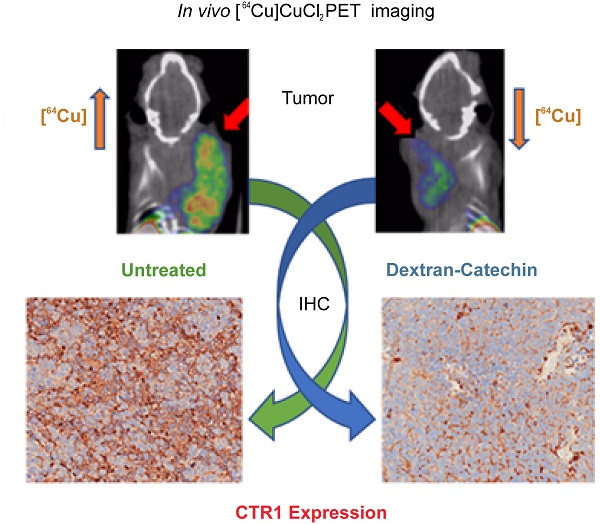
Given the strong clinical evidence that copper levels are significantly elevated in a wide spectrum of tumors, copper homeostasis is considered as an emerging target for anticancer drug design. Monitoring copper levels in vivo is therefore of paramount importance when assessing the efficacy of copper-targeting drugs. Herein, we investigated the activity of the copper-targeting compound Dextran-Catechin by developing a [64Cu]CuCl2 PET imaging protocol to monitor its effect on copper homeostasis in tumors.
Methods: Protein expression of copper transporter 1 (CTR1) in tissue microarrays representing 90 neuroblastoma patient tumors was assessed by immunohistochemistry. Western blotting analysis was used to study the effect of Dextran-Catechin on the expression of CTR1 in neuroblastoma cell lines and in tumors. A preclinical human neuroblastoma xenograft model was used to study anticancer activity of Dextran-Catechin in vivo and its effect on tumor copper homeostasis. PET imaging with [64Cu]CuCl2 was performed in such preclinical neuroblastoma model to monitor alteration of copper levels in tumors during treatment.
Results: CTR1 protein was found to be highly expressed in patient neuroblastoma tumors by immunohistochemistry. Treatment of neuroblastoma cell lines with Dextran-Catechin resulted in decreased levels of glutathione and in downregulation of CTR1 expression, which caused a significant decrease of intracellular copper. No changes in CTR1 expression was observed in normal human astrocytes after Dextran-Catechin treatment. In vivo studies and PET imaging analysis using the neuroblastoma preclinical model revealed elevated [64Cu]CuCl2 retention in the tumor mass. Following treatment with Dextran-Catechin, there was a significant reduction in radioactive uptake, as well as reduced tumor growth. Ex vivo analysis of tumors collected from Dextran-Catechin treated mice confirmed the reduced levels of CTR1. Interestingly, copper levels in blood were not affected by treatment, demonstrating potential tumor specificity of Dextran-Catechin activity.
Conclusion: Dextran-Catechin mediates its activity by lowering CTR1 and intracellular copper levels in tumors. This finding further reveals a potential therapeutic strategy for targeting copper-dependent cancers and presents a novel PET imaging method to assess patient response to copper-targeting anticancer treatments.
Keywords: Copper, Anticancer drugs, PET, Neuroblastoma, Molecular imaging
 Global reach, higher impact
Global reach, higher impact