13.3
Impact Factor
Theranostics 2018; 8(20):5519-5528. doi:10.7150/thno.27674 This issue Cite
Research Paper
Injection of Adipose-Derived Stromal Cells in the Knee of Patients with Severe Osteoarthritis has a Systemic Effect and Promotes an Anti-Inflammatory Phenotype of Circulating Immune Cells
1. Clinical Immunology and Osteoarticular Diseases Therapeutic Unit, Department of Rheumatology, Lapeyronie University Hospital, Montpellier, France.
2. IRMB, INSERM, Université Montpellier, Montpellier, France.
3. Department of Rheumatology, School of Medicine, University of Los Andes, Santiago, Chile.
4. University Hospital Wuerzburg, Department of Tissue Engineering and Regenerative Medicine, Wuerzburg, Germany.
5. Fraunhofer Institute for Silicate Research ISC, Translational Center Regenerative Therapies, Wuerzburg, Germany.
6. Department of Orthopedic and Trauma Surgery, Evangelisches Waldkrankenhaus Spandau, Berlin, Germany.
*these authors contributed equally to this work.
Abstract
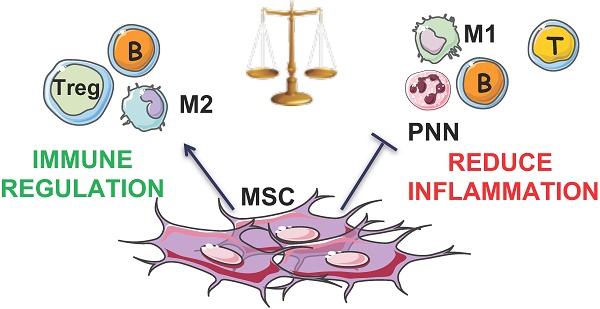
Rationale: Recent studies confirmed that osteoarthritis (OA) is associated with systemic inflammation. Adipose-derived stromal cells (ASCs) could become the most promising cell-based therapy in OA, based not only on their differentiation capacities and trophic and paracrine effects on the existing cartilage, but also on their immunomodulatory properties. Here, we wanted to determine the biological effect of autologous ASC intra-articular (IA) injection.
Method: To this aim, we monitored the profile of immune cells in fresh peripheral blood after IA injection of autologous ASCs in the knee of 18 patients with severe OA (ADIPOA phase I study). Specifically, we used 8-color flow cytometry antibody panels to characterize the frequencies of innate and adaptive immune cell subsets (monocytes, dendritic cells, regulatory T cells and B cells) in blood samples at baseline (before injection) and one week, one month and three months after ASC injection.
Results: We found that the percentage of CD4+CD25highCD127lowFOXP3+ regulatory T cells was significantly increased at 1 month after ASC injection, and this effect persisted for at least 3 months. Moreover, CD24highCD38high transitional B cells also were increased, whereas the percentage of classical CD14+ monocytes was decreased, at 3 months after ASC injection. These results suggest a global switch toward regulatory immune cells following IA injection of ASCs, underscoring the safety of ASC-based therapy. We did not find any correlation between the scores for the Visual Analogic Scale for pain, the Western Ontario and McMaster Universities Osteoarthritis Index (pain subscale and total score) at baseline and the immune cell profile changes, but this could be due to the small number of analyzed patients.
Conclusion: ASCs may drive an immediate local response by releasing paracrine factors and cytokines, and our results suggest that ASCs could also initiate a cascade resulting in a long-lasting systemic immune modulation.
Keywords: osteoarthritis, adipose-derived stromal cells, mesenchymal stromal cells, immune monitoring, multiparametric flow cytometry, immunomodulation
 Global reach, higher impact
Global reach, higher impact