Impact Factor
Theranostics 2018; 8(19):5501-5518. doi:10.7150/thno.27721 This issue Cite
Research Paper
Self-assembly of porphyrin-grafted lipid into nanoparticles encapsulating doxorubicin for synergistic chemo-photodynamic therapy and fluorescence imaging
1. Department of Biomedical Engineering, College of Engineering, Peking University, Beijing 100871, China
2. Department of Ultrasonography, Peking University Third Hospital, Beijing 100191, China
*These authors contributed equally to this work.
Abstract
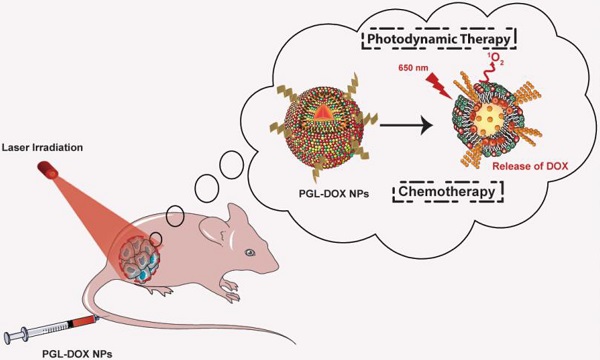
The limited clinical efficacy of monotherapies in the clinic has urged the development of novel combination platforms. Taking advantage of light-triggered photodynamic treatment combined together with the controlled release of nanomedicine, it has been possible to treat cancer without eliciting any adverse effects. However, the challenges imposed by limited drug loading capacity and complex synthesis process of organic nanoparticles (NPs) have seriously impeded advances in chemo-photodynamic combination therapy. In this experiment, we utilize our previously synthesized porphyrin-grafted lipid (PGL) NPs to load highly effective chemotherapeutic drug, doxorubicin (DOX) for synergistic chemo-photodynamic therapy.
Methods: A relatively simple and inexpensive rapid injection method was used to prepare porphyrin-grafted lipid (PGL) NPs. The self-assembled PGL NPs were used further to encapsulate DOX via a pH-gradient loading protocol. The self-assembled liposome-like PGL NPs having a hydrophilic core were optimized to load DOX at an encapsulation efficiency (EE) of ~99%. The resultant PGL-DOX NPs were intact, highly stable and importantly these NPs successfully escaped from the endo-lysosomal compartment after laser irradiation to release DOX in the cytosol. The therapeutic efficacy of the aforementioned formulation was validated both in vitro and in vivo.
Results: PGL-DOX NPs demonstrated excellent cellular uptake, chemo-photodynamic response, and fluorescence imaging ability in different cell lines. Under laser irradiation, cells treated with a low molar concentration of PGL-DOX NPs reduced cell viability significantly. Moreover, in vivo experiments conducted in a xenograft mouse model further demonstrated the excellent tumor accumulation capability of PGL-DOX NPs driven by the enhanced permeability and retention (EPR) effect. Through fluorescence imaging, the biodistribution of PGL-DOX NPs in tumor and major organs was also easily monitored in real time in vivo. The inherent ability of porphyrin to generate ROS under laser irradiation combined with the cytotoxic effect of the anticancer drug DOX significantly suppressed tumor growth in vivo.
Conclusion: In summary, the PGL-DOX NPs combined chemo-photodynamic nanoplatform may serve as a potential candidate for cancer theranostics.
Keywords: porphyrin, doxorubicin, theranostics, chemotherapy, photodynamic therapy
 Global reach, higher impact
Global reach, higher impact