Impact Factor
Theranostics 2018; 8(19):5434-5451. doi:10.7150/thno.27882 This issue Cite
Research Paper
Microglial TLR4-dependent autophagy induces ischemic white matter damage via STAT1/6 pathway
1. Department of Neurology, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430030, China
2. Department of Neurology, Mayo Clinic, Rochester, MN 55905
* These two authors contributed equally to this work.
Abstract
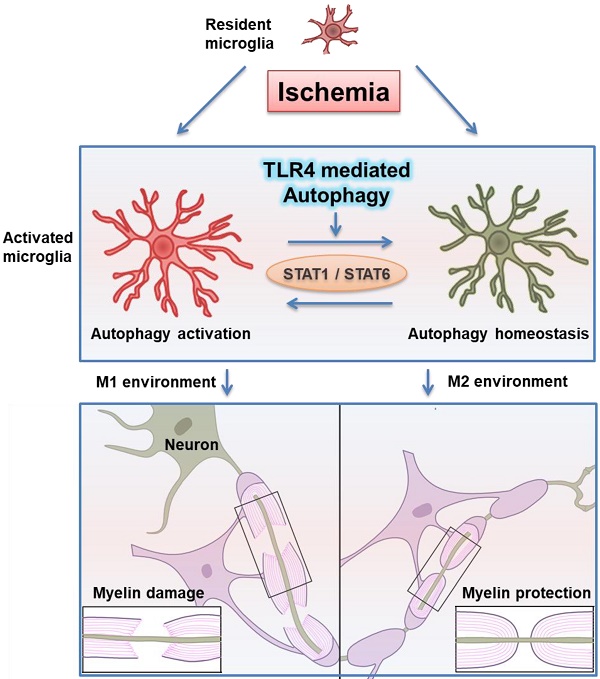
Rationale: Ischemic white matter damage frequently results in myelin loss, accompanied with microglial activation. We previously found that directing microglia towards an anti-inflammatory phenotype provided a beneficial microenvironment and helped maintain white matter integrity during chronic cerebral hypoperfusion. However, the molecular mechanisms underlying microglial polarization remain elusive.
Methods: Hypoperfusion induced white matter damage mice model and lipopolysaccharide (LPS) induced primary cultured microglia were established. Autophagy activation in microglia was detected both in vivo and in vitro by immunofluorescence, Western blot and electron microscopy. Autophagy inhibitors/agonist were administrated to investigate the role of autophagic process in modulating microglial phenotypes. Quantitative real time-polymerase chain reaction and Western blot were carried out to investigate the possible pathway.
Results: We identified rapid accumulation of autophagosomes in primary cultured microglia exposed to LPS and within activated microglia during white matter ischemic damage. Autophagy inhibitors switched microglial function from pro-inflammatory to anti-inflammatory phenotype. Furthermore, we found TLR4, one of the major receptors binding LPS, was most highly expressed on microglia in corpus callosum during white matter ischemic damage, and TLR4 deficiency could mimic the phenomenon in microglial functional transformation, and exhibit a protective activity in chronic cerebral hypoperfusion. Whereas, the anti-inflammatory phenotype of microglia in TLR4 deficiency group was largely abolished by the activation of autophagic process. Finally, our transcriptional analysis confirmed that the up-regulation of STAT1 and down-regulation of STAT6 in microglia exposure to LPS could be reversed by autophagy inhibition.
Conclusion: These results indicated that TLR4-dependent autophagy regulates microglial polarization and induces ischemic white matter damage via STAT1/6 pathway.
Keywords: TLR4, autophagy, white matter ischemic injury, microglia, STAT
 Global reach, higher impact
Global reach, higher impact