13.3
Impact Factor
Theranostics 2018; 8(18):4912-4924. doi:10.7150/thno.27608 This issue Cite
Research Paper
Blood exosomes regulate the tissue distribution of grapefruit-derived nanovector via CD36 and IGFR1 pathways
1. Robley Rex VA Medical Center, Louisville, KY 40206
2. James Graham Brown Cancer Center, Department of Microbiology & Immunology, University of Louisville, KY 40202
3. Department of Medicine, University of Louisville, KY 40202
4. Department of Central Laboratory and Huai'an Key Laboratory of Esophageal Cancer Biobank, The Affiliated Huaian No.1 People's Hospital of Nanjing Medical University, Huai'an 223300, China.
# These authors contributed equally to this work
Abstract
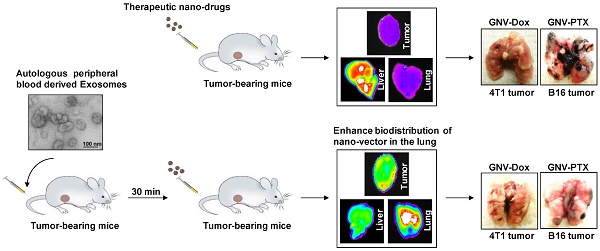
Tumor-specific delivery of therapeutics is challenging. One of the major hurdles for successfully delivering targeted agents by nanovectors is the filtering role of the liver in rapidly sequestering nanovectors from the circulation. Exosomes, a type of endogenous nanoparticle, circulate continuously in the peripheral blood and play a role in intercellular communication. The aim of this study was to determine whether the level of endogenous exosomes has an effect on nanovector delivery efficiency of targeted agents.
Methods: Exosomes were isolated from peripheral blood and intravenously (I.V.) injected into tumor-bearing mice. Subsequently, 1,1-dioctadecyl-3,3,3'3'-tetramethylindotricarbocyanine-iodide (DiR) fluorescent dye-labeled nanoparticles, including grapefruit nanovectors (GNV) and standard liposomes, were I.V. injected in the mice. The efficiency of redirecting GNVs from liver to other organs of injected mice was further analyzed with in vivo imaging. The concentration of chemo drugs delivered by GNV was measured by HPLC and the anti-lung metastasis therapeutic effects of chemo drugs delivered by GNVs in mouse breast cancer and melanoma cancer models were evaluated.
Results: We show that tail vein-injected exosomes isolated from mouse peripheral blood were predominately taken up by liver Kupffer cells. Injection of peripheral blood-derived exosomes before I.V. injection of grapefruit-derived nanovector (GNV) decreased the deposition of GNV in the liver and redirected the GNV to the lung and to the tumor in breast and melanoma tumor-bearing mouse models. Enhanced therapeutic efficiency of doxorubicin (Dox) or paclitaxel (PTX) carried by GNVs for lung metastases was demonstrated when there was an I.V. injection of exosomes before therapeutic treatment. Furthermore, we found that CD36 and IGFR1 receptor-mediated pathways played a critical role in the exosome-mediated inhibitory effect of GNV entry into liver macrophages.
Conclusions: Collectively, our findings provide a foundation for using autologous exosomes to enhance therapeutic vector targeted delivery to the lung.
Keywords: Blood exosomes, nanovector uptake, liver Kupffer cells, CD36 and IGFR1, outer nuclear membrane cluster, lung metastasis
 Global reach, higher impact
Global reach, higher impact