13.3
Impact Factor
Theranostics 2018; 8(16):4477-4490. doi:10.7150/thno.26249 This issue Cite
Research Paper
Integration of magnetic resonance imaging and protein and metabolite CSF measurements to enable early diagnosis of secondary progressive multiple sclerosis
1. Department of Medical Sciences, Clinical Chemistry, Uppsala University, Sweden
2. Department of Pharmaceutical Biosciences, Uppsala University, Sweden
3. Department of Neuroscience, Uppsala University, Sweden
4. Unit of Rheumatology, Department of Medicine, Karolinska Institutet, Stockholm, Sweden
5. Department of Psychiatry and Neurochemistry, the Sahlgrenska Academy at the University of Gothenburg, Mölndal, Sweden
6. Clinical Neurochemistry Laboratory, Sahlgrenska University Hospital, Mölndal, Sweden
7. UCL Institute of Neurology, Queen Square, London, United Kingdom
8. UK Dementia Research Institute at UCL, London, United Kingdom
Abstract
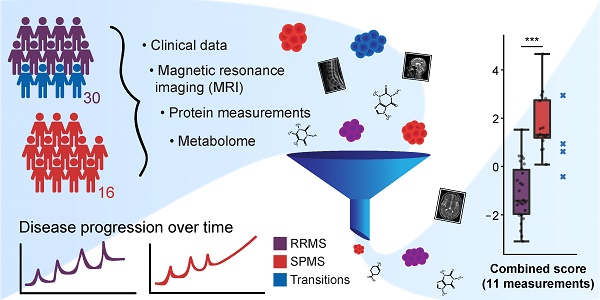
Molecular networks in neurological diseases are complex. Despite this fact, contemporary biomarkers are in most cases interpreted in isolation, leading to a significant loss of information and power. We present an analytical approach to scrutinize and combine information from biomarkers originating from multiple sources with the aim of discovering a condensed set of biomarkers that in combination could distinguish the progressive degenerative phenotype of multiple sclerosis (SPMS) from the relapsing-remitting phenotype (RRMS).
Methods: Clinical and magnetic resonance imaging (MRI) data were integrated with data from protein and metabolite measurements of cerebrospinal fluid, and a method was developed to sift through all the variables to establish a small set of highly informative measurements. This prospective study included 16 SPMS patients, 30 RRMS patients and 10 controls. Protein concentrations were quantitated with multiplexed fluorescent bead-based immunoassays and ELISA. The metabolome was recorded using liquid chromatography-mass spectrometry. Clinical follow-up data of the SPMS patients were used to assess disease progression and development of disability.
Results: Eleven variables were in combination able to distinguish SPMS from RRMS patients with high confidence superior to any single measurement. The identified variables consisted of three MRI variables: the size of the spinal cord and the third ventricle and the total number of T1 hypointense lesions; six proteins: galectin-9, monocyte chemoattractant protein-1 (MCP-1), transforming growth factor alpha (TGF-α), tumor necrosis factor alpha (TNF-α), soluble CD40L (sCD40L) and platelet-derived growth factor AA (PDGF-AA); and two metabolites: 20β-dihydrocortisol (20β-DHF) and indolepyruvate.
The proteins myelin basic protein (MBP) and macrophage-derived chemokine (MDC), as well as the metabolites 20β-DHF and 5,6-dihydroxyprostaglandin F1a (5,6-DH-PGF1), were identified as potential biomarkers of disability progression.
Conclusion: Our study demonstrates, in a limited but well-defined and data-rich cohort, the importance and value of combining multiple biomarkers to aid diagnostics and track disease progression.
Keywords: data integration, multiple sclerosis, disease progression, metabolomics, biomarker
 Global reach, higher impact
Global reach, higher impact