Impact Factor
Theranostics 2018; 8(16):4462-4476. doi:10.7150/thno.24395 This issue Cite
Research Paper
Radionuclide imaging of VEGFR2 in glioma vasculature using biparatopic affibody conjugate: proof-of-principle in a murine model
1. Department of Medicinal Chemistry, Uppsala University, Sweden;
2. Department of Protein Science, School of Engineering Sciences in Chemistry, Biotechnology and Health, KTH Royal Institute of Technology, Sweden;
3. Department of Immunology, Genetics and Pathology, Uppsala University, Sweden
4. Preclinical PET-MRI Platform, Department of Medicinal Chemistry, Uppsala University, Sweden
5. Science for Life Laboratory, Uppsala University, Sweden
* These authors contributed equally
Abstract
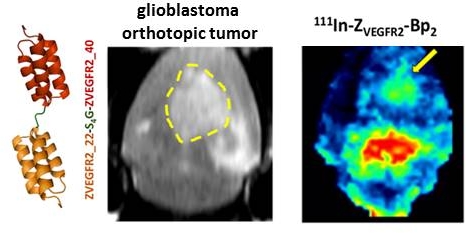
Vascular endothelial growth factor receptor-2 (VEGFR2) is a key mediator of angiogenesis and therefore a promising therapeutic target in malignancies including glioblastoma multiforme (GBM). Molecular imaging of VEGFR2 expression may enable patient stratification for antiangiogenic therapy. The goal of the current study was to evaluate the capacity of the novel anti-VEGFR2 biparatopic affibody conjugate (ZVEGFR2-Bp2) for in vivo visualization of VEGFR2 expression in GBM.
Methods: ZVEGFR2-Bp2 coupled to a NODAGA chelator was generated and radiolabeled with indium-111. The VEGFR2-expressing murine endothelial cell line MS1 was used to evaluate in vitro binding specificity and affinity, cellular processing and targeting specificity in mice. Further tumor targeting was studied in vivo in GL261 glioblastoma orthotopic tumors. Experimental imaging was performed.
Results: [111In]In-NODAGA-ZVEGFR2-Bp2 bound specifically to VEGFR2 (KD=33±18 pM). VEGFR2-mediated accumulation was observed in liver, spleen and lungs. The tumor-to-organ ratios 2 h post injection for mice bearing MS1 tumors were approximately 11 for blood, 15 for muscles and 78 for brain. Intracranial GL261 glioblastoma was visualized using SPECT/CT. The activity uptake in tumors was significantly higher than in normal brain tissue. The tumor-to-cerebellum ratios after injection of 4 µg [111In]In-NODAGA-ZVEGFR2-Bp2 were significantly higher than the ratios observed for the 40 µg injected dose and for the non-VEGFR2 binding size-matched conjugate, demonstrating target specificity. Microautoradiography of cryosectioned CNS tissue was in good agreement with the SPECT/CT images.
Conclusion: The anti-VEGFR2 affibody conjugate [111In]In-NODAGA-ZVEGFR2-Bp2 specifically targeted VEGFR2 in vivo and visualized its expression in a murine GBM orthotopic model. Tumor-to-blood ratios for [111In]In-NODAGA-ZVEGFR2-Bp2 were higher compared to other VEGFR2 imaging probes. [111In]In-NODAGA-ZVEGFR2-Bp2 appears to be a promising probe for in vivo noninvasive visualization of tumor angiogenesis in glioblastoma.
Keywords: VEGFR2, affibody molecule, molecular imaging, SPECT, orthotopic glioma model, in vivo
 Global reach, higher impact
Global reach, higher impact