13.3
Impact Factor
Theranostics 2018; 8(16):4393-4408. doi:10.7150/thno.26869 This issue Cite
Review
State-of-the-art of microbubble-assisted blood-brain barrier disruption
1. Department of Mechanical Engineering, University of Colorado, Boulder, CO 80309
2. Intramural Research Program, National Institute on Drug Abuse, Baltimore, MD 21224
Abstract
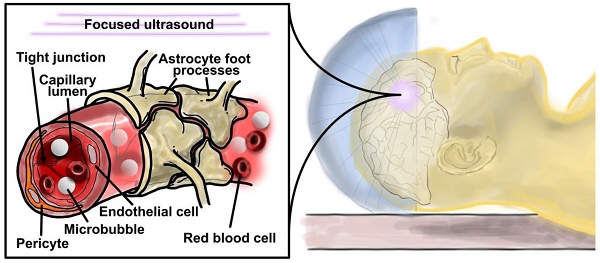
Focused ultrasound with microbubbles promises unprecedented advantages for blood-brain barrier disruption over existing intracranial drug delivery methods, as well as a significant number of tunable parameters that affect its safety and efficacy. This review provides an engineering perspective on the state-of-the-art of the technology, considering the mechanism of action, effects of microbubble properties, ultrasound parameters and physiological variables, as well as safety and potential therapeutic applications. Emphasis is placed on the use of unified parameters, such as microbubble volume dose (MVD) and ultrasound mechanical index, to optimize the procedure and establish safety limits. It is concluded that, while efficacy has been demonstrated in several animal models with a wide range of payloads, acceptable measures of safety should be adopted to accelerate collaboration and improve understanding and clinical relevance.
Keywords: focused ultrasound, ultrasound contrast agent, drug delivery, gene therapy
 Global reach, higher impact
Global reach, higher impact