Impact Factor
Theranostics 2018; 8(16):4372-4392. doi:10.7150/thno.27088 This issue Cite
Research Paper
3D printing of Mo-containing scaffolds with activated anabolic responses and bi-lineage bioactivities
1. State Key Laboratory of High Performance Ceramics and Superfine Microstructure, Shanghai Institute of Ceramics, Chinese Academy of Sciences, Shanghai 200050, People's Republic of China.
2. University of Chinese academy of Sciences, Beijing, People's Republic of China
3. Department of Orthopaedic Surgery Digital Medicine Institute, Nanjing Medical University, Nanjing Hospital. No. 68 Changle Road Nanjing, 210006, People's Republic of China
W. Dang and X.Wang are the co-first authors.
Abstract
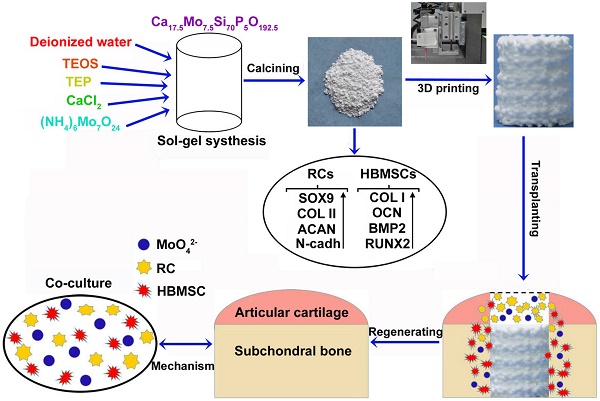
When osteochondral tissues suffer from focal or degenerative lesions caused by trauma or disorders, it is a tough challenge to regenerate them because of the limited self-healing capacity of articular cartilage. In this study, a series of Mo-doped bioactive glass ceramic (Mo-BGC) scaffolds were prepared and then systematically characterized. The released MoO42- ions from 7.5Mo-BGC scaffolds played a vital role in regenerating articular cartilage and subchondral bone synchronously.
Methods: The Mo-BGC scaffolds were fabricated through employing both a sol-gel method and 3D printing technology. SEM, EDS, HRTEM, XRD, ICPAES and mechanical strength tests were respectively applied to analyze the physicochemical properties of Mo-BGC scaffolds. The proliferation and differentiation of rabbit chondrocytes (RCs) and human bone mesenchymal stem cells (HBMSCs) cultured with dilute solutions of 7.5Mo-BGC powder extract were investigated in vitro. The co-culture model was established to explore the possible mechanism of stimulatory effects of MoO42- ions on the RCs and HBMSCs. The efficacy of regenerating articular cartilage and subchondral bone using 7.5Mo-BGC scaffolds was evaluated in vivo.
Results: The incorporation of Mo into BGC scaffolds effectively enhanced the compressive strength of scaffolds owing to the improved surface densification. The MoO42- ions released from the 7.5Mo-BGC powders remarkably promoted the proliferation and differentiation of both RCs and HBMSCs. The MoO42- ions in the co-culture system significantly stimulated the chondrogenic differentiation of RCs and meanwhile induced the chondrogenesis of HBMSCs. The chondrogenesis stimulated by MoO42- ions happened through two pathways: 1) MoO42- ions elicited anabolic responses through activating the HIF-1α signaling pathway; 2) MoO42- ions inhibited catabolic responses and protected cartilage matrix from degradation. The in vivo study showed that 7.5Mo-BGC scaffolds were able to significantly promote cartilage/bone regeneration when implanted into rabbit osteochondral defects for 8 and 12 weeks, displaying bi-lineage bioactivities.
Conclusion: The 3D-printed Mo-BGC scaffolds with bi-lineage bioactivities and activated anabolic responses could offer an effective strategy for cartilage/bone interface regeneration.
 Global reach, higher impact
Global reach, higher impact