Impact Factor
Theranostics 2017; 7(18):4537-4550. doi:10.7150/thno.19546 This issue Cite
Research Paper
pH-Responsive Aerobic Nanoparticles for Effective Photodynamic Therapy
1. Department of Oral & Maxillofacial-Head & Neck Oncology, Shanghai Key Laboratory of Stomatology Cosmetic Laser Center, Ninth People's Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai 200011, P. R. China.
2. School of Chemistry and Chemical Engineering, Shanghai Key Lab of Electrical Insulation and Thermal Aging, Shanghai Jiao Tong University, 800 Dongchuan Road, Shanghai, 200240, P. R. China.
3. School of Chemical and Environmental Engineering, Shanghai Institute of Technology, 100 Haiquan Road, Shanghai 201418, P. R. China.
Abstract
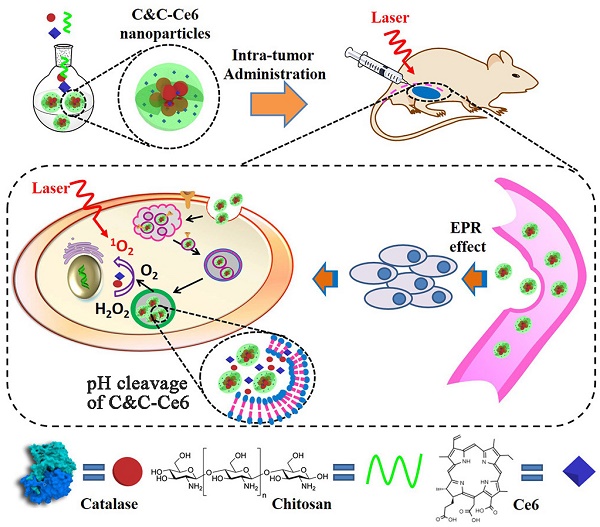
Rationale: Photodynamic therapy (PDT), an O2-dependent treatment for inhibition of cancer proliferation, suffers from the low therapeutic effect in clinical application due to the hypoxic microenvironment in tumor cells.
Methods: To overcome this obstacle, a stimuli-responsive drug delivery system with O2 self-sufficiency for effective PDT was developed. In this study, pH-responsive aerobic nanoparticles were prepared by the electrostatic interaction between the O2-evolving protein Catalase and Chitosan. Subsequently, the photosensitizer Chlorin e6 (Ce6) was encapsulated in the nanoparticles.
Results: The nanoparticles exhibited high stability in aqueous medium and efficient cellular uptake by tumor cells facilitating their accumulation in tumors by enhanced permeability and retention (EPR) effect. In acidic environment, irradiation caused disassembly of the nanoparticles resulting in the quick release of Catalase and the photosensitizer with continuous formation of cytotoxic singlet oxygen (1O2) greatly enhancing the PDT efficacy in hypoxic tumor tissues both in vitro and in vivo biological studies.
Conclusion: Due to the unique O2 self-sufficiency, the nanoparticles, upon irradiation, exhibited higher anticancer activity than free Ce6 both in vitro and in vivo. Our work has identified a new pH-triggered strategy to overcome hypoxia for effective PDT against cancer cells.
Keywords: pH-Responsive, Nanoparticles, Catalase, Hydrogen Peroxide, Photodynamic Therapy
 Global reach, higher impact
Global reach, higher impact