13.3
Impact Factor
Theranostics 2019; 9(5):1510-1522. doi:10.7150/thno.29620 This issue Cite
Research Paper
p66Shc Contributes to Liver Fibrosis through the Regulation of Mitochondrial Reactive Oxygen Species
1. Department of Pharmacology, Dalian Medical University, Dalian 116044, China.
2. Department of Surgery, The Second Affiliated Hospital of Dalian Medical University, Dalian 116023, China.
3. Department of Pharmacy, The Second Affiliated Hospital of Dalian Medical University, Dalian 116023, China.
4. Department of Pharmacy, The First Affiliated Hospital of Dalian Medical University, Dalian 116011, China.
*These authors contributed equally to this work.
Abstract
Background: p66Shc is a redox enzyme that mediates mitochondrial reactive oxygen species (ROS) generation. p66Shc inhibition confers protection against liver injury, however, its functional contribution to liver fibrosis remains unclear. The aim of this study is to explore the involvement of p66Shc in liver fibrosis and underlying mechanism of p66Shc by focusing on mitochondrial ROS.
Methods: p66Shc-silenced mice were injected with carbon tetrachloride (CCl4). Primary hepatic stellate cells (HSCs) were performed with p66Shc silencing or overexpression prior to TGF-β1 stimulation.
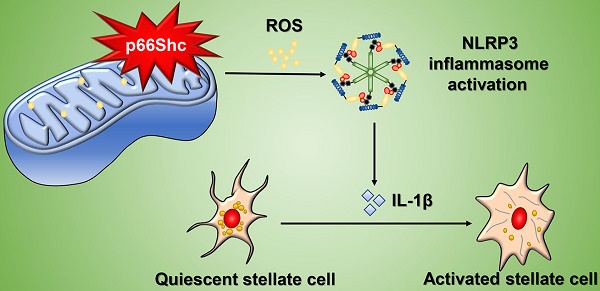
Results: p66Shc expression was progressively elevated in mice with CCl4-induced liver fibrosis, and p66Shc silencing in vivo significantly attenuated fibrosis development, reducing liver damage, oxidative stress and HSC activation, indicated by the decreased α-SMA, CTGF and TIMP1 levels. Furthermore, in primary HSCs, p66Shc-mediated mitochondrial ROS production played a vital role in mitochondrial morphology and cellular metabolism. Knockdown of p66Shc significantly inhibited mitochondrial ROS production and NOD-like receptor protein 3 (NLRP3) inflammasome activation, which were closely associated with HSC activation, indicated by the decreased α-SMA, CTGF and TIMP1 levels. However, p66Shc overexpression exerted the opposite effects, which were suppressed by a specific mitochondrial ROS scavenger (mito-TEMPO). More importantly, p66Shc expression was significantly increased in human with liver fibrosis, accompanied by NLRP3 inflammasome activation.
Conclusions: p66Shc is a key regulator of liver fibrosis by mediating mitochondrial ROS production, which triggers NLRP3 inflammasome activation.
Keywords: p66Shc, mitochondrial reactive oxygen species, liver fibrosis, hepatic stellate cell, NLRP3 inflammasome
 Global reach, higher impact
Global reach, higher impact