Impact Factor
Theranostics 2018; 8(16):4359-4371. doi:10.7150/thno.26900 This issue Cite
Research Paper
Inactivation of PBX3 and HOXA9 by down-regulating H3K79 methylation represses NPM1-mutated leukemic cell survival
1. State Key Laboratory for Medical Genomics, Shanghai Institute of Hematology, Rui-Jin Hospital affiliated to Shanghai Jiao-Tong University School of Medicine, Shanghai, China
2. Department of Hematology, the First Hospital of China Medical University, Shenyang, China
3. Division of Genetics, Department of Medicine, Beth Israel Deaconess Medical Center, Harvard Medical School, Boston, MA, USA
4. Department of Hematology, Renmin Hospital of Wuhan University, Wuhan, Hubei, China
# W.Z., C.Z. and J.M.Z. made equal contribution.
Abstract
Acute myeloid leukemia (AML) with an NPM1 mutation (NPMc+) has a distinct gene expression signature and displays molecular abnormalities similar to mixed lineage leukemia (MLL), including aberrant expression of the PBX3 and HOXA gene cluster. However, it is unclear if the aberrant expression of PBX3 and HOXA is essential for the survival of NPM1-mutated leukemic cells.
Methods: Using the gene expression profiling of TCGA and E-MTAB-3444 datasets, we screened for high co-expression of PBX3 and HOXA9 in NPMc+ leukemia patients. We performed NPMc+ depletion and overexpression experiments to examine aberrant H3K79 methylation through epigenetic regulation. Through RNA interference technology and small-molecule inhibitor treatment, we evaluated the effect of methyl-modified H3K79 on cell survival and explored the possible underlying mechanism.
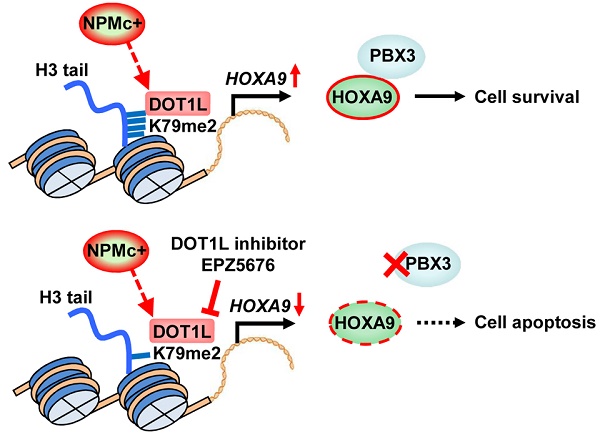
Results: We showed that NPMc+ increased the expression of PBX3 and HOXA9, which are both poor prognosis indicators in AML. High PBX3 and HOXA9 expression was accompanied by increased dimethylated and trimethylated H3K79 in transgenic murine Lin-Sca-1+c-Kit+ cells and human NPMc+ leukemia cells. Using chromatin immunoprecipitation sequencing (ChIP-seq) assays of NPMc+ cells, we determined that hypermethylated H3K79 was present at the expressed HOXA9 gene but not the PBX3 gene. PBX3 expression was positively regulated by HOXA9, and a reduction in either PBX3 or HOXA9 resulted in NPMc+ cell apoptosis. Importantly, an inhibitor of DOT1L, EPZ5676, effectively and selectively promoted NPMc+ human leukemic cell apoptosis by reducing HOXA9 and PBX3 expression.
Conclusion: Our data indicate that NPMc+ leukemic cell survival requires upregulation of PBX3 and HOXA9, and this action can be largely attenuated by a DOT1L inhibitor.
Keywords: NPM1 mutation, H3K79 hypermethylation, PBX3, HOXA9, DOT1L inhibitor
 Global reach, higher impact
Global reach, higher impact